Product Quality Reviews (PQRs) serve as a cornerstone in GMP, comprehensively evaluating production and quality control processes. This systematic review confirms product quality and consistency and identifies areas for improvement, ensuring that every batch produced meets the highest standards.
In this article, we will look into the essential components, objectives, and challenges of PQR, highlighting its critical role in maintaining the integrity of pharmaceutical products.
A Product Quality Review (PQR) is a comprehensive evaluation of the production and control of a pharmaceutical product. It is designed to verify the consistency of the process, identify any trends, and ensure that the product remains in compliance with regulatory standards.
PQRs are essential for maintaining the integrity and safety of pharmaceutical products, providing a detailed review of every aspect of production from raw materials to finished products.
RELATED: Different Types of GMP Documentation Used In Pharma Industry
The primary purpose of a PQR is to systematically evaluate the quality of a pharmaceutical product over a specified period. This ongoing assessment ensures that the manufacturing processes consistently produce products that meet predefined quality standards and regulatory requirements. By reviewing data from various batches, PQR aims to achieve several critical objectives:
The frequency of conducting Product Quality Reviews (PQRs) is typically on an annual basis. This annual frequency is a regulatory requirement set by major health authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Conducting PQRs annually ensures continuous monitoring and improvement of product quality, which is essential for maintaining the integrity and safety of pharmaceutical products.
According to the FDA, as specified in 21 CFR Part 211.180(e), pharmaceutical companies are required to conduct an Annual Product Review (APR). This review must include an evaluation of all batches produced and highlight any trends or deviations that could impact product quality.
The annual review helps ensure that the product consistently meets quality standards and identifies any necessary adjustments or improvements in the manufacturing process.
The EMA outlines its requirements for Product Quality Reviews in Chapter 1 of EudraLex Volume 4, specifically in Section 1.10. This section mandates that marketing authorization holders conduct an annual PQR. The review should cover all batches released, returned, or rejected and evaluate any deviations or changes that could affect product quality.
This annual review is crucial for maintaining compliance with EMA regulations and ensuring that the product remains safe and effective for patients.
If no batches of a particular product are manufactured during a given year, it is still essential to conduct a PQR. The focus of the PQR in such cases will be on confirming that there were no production activities and ensuring that all systems remain ready for future manufacturing. This includes:
RELATED: Supplier Qualification in GMP
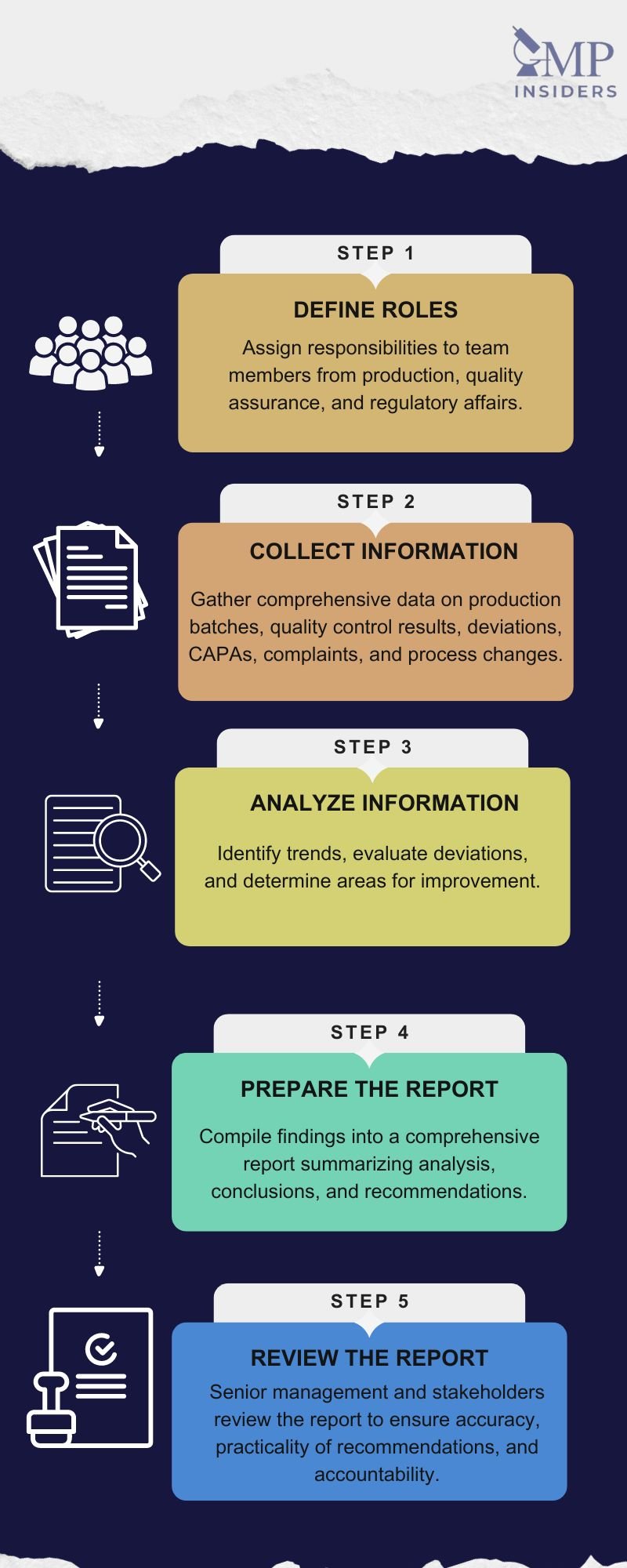
The Product Quality Review (PQR) process involves several critical stages to ensure a comprehensive evaluation of the product’s quality over time. Each stage is essential for a thorough and effective review. Below are the key stages, along with their roles and importance.
The first stage of the PQR process is to define roles and assign responsibilities to team members. This includes personnel from production, quality assurance, and regulatory affairs. Clearly defined roles ensure that every aspect of the PQR is covered efficiently and that there is accountability for each task. Team members need to understand their specific duties and how they contribute to the overall review process.
Once roles are defined, the next step is to collect information. This involves gathering comprehensive data on:
After collecting the necessary data, the next stage is to analyze the information in detail. This analysis aims to:
The findings from the data analysis are then compiled into a comprehensive report. This report should:
The final stage of the PQR process is the review of the report by senior management and relevant stakeholders. This review ensures that the report:
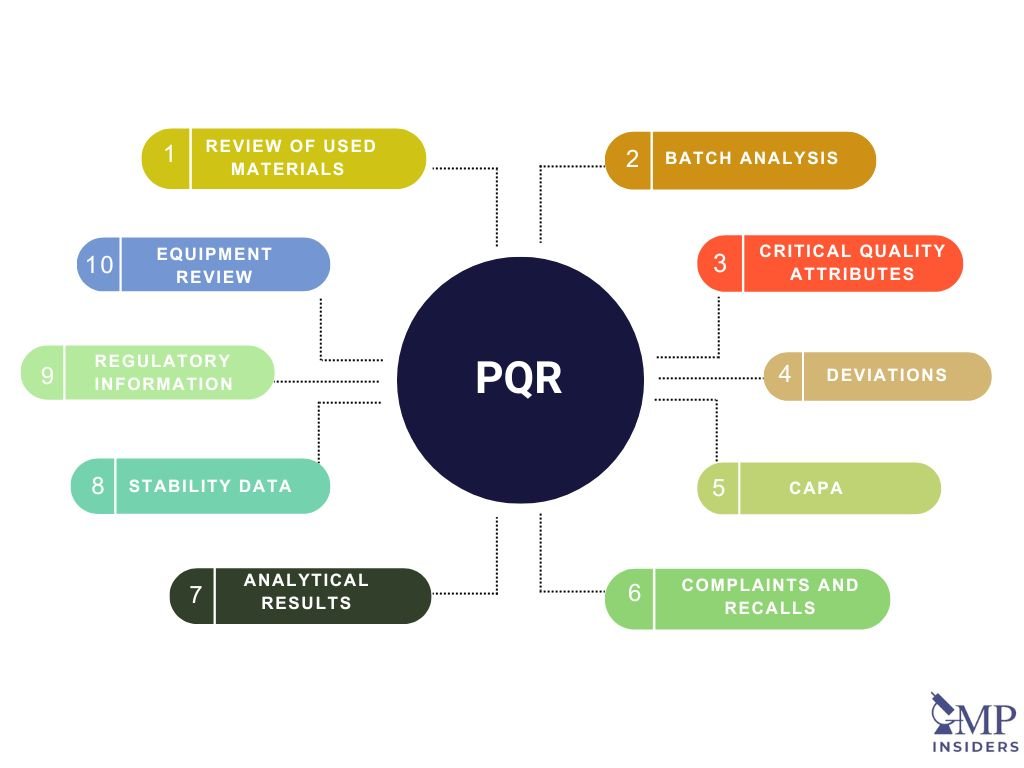
A thorough Product Quality Review (PQR) is a comprehensive evaluation that includes several critical components. These components ensure a detailed analysis of the production process, product quality, and any issues that may have arisen during the review period. The key elements of a PQR are as follows:
RELATED: Out of Specification (OOS) Results
RELATED: Corrective and Prevention Action (CAPA) Plan in GMP
Conducting a Product Quality Review (PQR) is essential for maintaining high standards of product quality and regulatory compliance in the pharmaceutical industry. However, this process comes with several challenges that companies must address to ensure a successful and thorough review. Here are some of the key challenges, along with practical tips for overcoming them.
One of the primary challenges in conducting a PQR is the efficient collection and management of data.
Challenges:
Tips:
After data collection, the next significant challenge is analyzing the data to identify trends and patterns.
Challenges:
Tips:
SEE ALSO: Out of Trend Results in Pharma Industry
Efficient resource allocation is crucial to the success of the PQR process.
Challenges:
Tips:
Staying compliant with regulatory requirements is another significant challenge in the PQR process.
Challenges:
Tips:
Effective communication is vital for the success of the PQR process and for implementing its findings.
Challenges:
Tips:
Data from a PQR is used to monitor product quality, identify trends, implement corrective actions, and ensure compliance with regulatory standards. It aids in continuous improvement and optimizing manufacturing processes.
PQR is a retrospective, comprehensive review of all production and quality data over a period, focusing on trends and overall performance. Quality Control Testing evaluates individual batches during production for compliance with quality specifications.
Yes, many aspects of PQR can be automated using specialized software tools that facilitate data collection, analysis, and reporting, improving efficiency and accuracy.
PQR helps identify potential quality issues early, allowing for proactive risk mitigation. By analyzing trends and deviations, it supports effective risk management and enhances product safety and compliance.
APQR (Annual Product Quality Review) is a specific type of PQR conducted annually to meet regulatory requirements. PQR is a broader term that may refer to periodic quality reviews conducted at different intervals depending on regulatory or internal requirements.
Product Quality Reviews (PQR) are indispensable for maintaining the high standards required in pharmaceutical manufacturing. By systematically evaluating production processes and quality attributes, PQRs ensure that each product batch meets predefined quality criteria and regulatory requirements.
Regularly conducting these reviews helps identify trends, optimize processes, and implement corrective actions, thereby safeguarding product quality and patient safety. As regulatory landscapes evolve, PQRs provide the necessary framework for continuous improvement and compliance, reinforcing the reliability and trust in pharmaceutical products.
Embracing robust PQR practices is not just a regulatory necessity but a fundamental aspect of delivering safe and effective pharmaceutical products to the market.