Moist heat sterilization remains the most widely used and trusted method in the pharmaceutical and bio-pharmaceutical industries. Despite its long history and broad regulatory acceptance, it remains one of the most misunderstood and inconsistently applied processes in GMP environments.
Many organizations continue to rely on legacy practices, most notably the default application of “121 °C for 15 minutes” without adequately considering load characteristics, air removal challenges, heat transfer mechanisms, or container integrity risks. This often results in inappropriate cycle selection, misaligned validation strategies, and avoidable GMP observations.
The last revision of EU GMP Annex 1 (2022) has fundamentally changed regulatory expectations. Manufacturers are now required to demonstrate scientific understanding, risk-based justification, and load-specific control of sterilization processes (Annex 1 §1.5, §2.1).
Moist heat sterilization is no longer viewed as a single, generic technology, but rather as a family of processes, each governed by different sterilization mechanisms, dominant risks, and acceptance criteria. These distinctions are expected to be clearly defined and justified within the firm’s Contamination Control Strategy (CCS).
This article introduces a practical, GMP-aligned risk-based decision tree for moist heat sterilization in the pharmaceutical industry. It is intended to support manufacturers in designing defensible sterilization strategies that meet current regulatory expectations, strengthen sterility assurance, and integrate seamlessly into the Pharmaceutical Quality System, including validation, change control, and contamination control strategy frameworks.
Fundamentals of Moist Heat Sterilization
Moist heat sterilization achieves microbial inactivation through the combined effects of saturated steam, temperature, and exposure time. The primary mechanism is the transfer of latent heat released during steam condensation on cooler surfaces, which leads to irreversible denaturation of proteins and enzymes within microorganisms. This mechanism is well understood, reproducible, and capable of achieving high levels of sterility assurance when properly controlled.

From a GMP perspective, the effectiveness of moist heat sterilization is not defined solely by reaching a target temperature. It depends on the ability of saturated steam to contact all critical load surfaces under conditions that allow efficient heat transfer. Any factor that interferes with steam contact or condensation directly reduces lethality and represents a sterilization risk.
Key lethality concepts underpinning moist heat sterilization include:
- F₀ value, which represents the equivalent exposure time at 121 °C delivering a defined level of microbial lethality.
- D-value, indicating the time required at a specific temperature to reduce a microbial population by one logarithmic unit.
- z-value, describing the temperature change needed to achieve a tenfold change in the D-value.
While these parameters provide a quantitative basis for sterilization, they do not, on their own, guarantee effective sterilization. The dominant risks in moist heat processes are typically physical rather than microbiological, and include:
- Incomplete air removal, preventing steam penetration
- Non-uniform heat distribution within the chamber or load
- Load shielding and cold spot formation
- Inadequate condensate removal
- Load configuration and packaging effects on heat transfer
These risks explain why identical time–temperature profiles can produce very different sterilization outcomes depending on load type, container system, and cycle design. Consequently, understanding the fundamentals of steam behavior, heat transfer, and physical constraints is essential before applying any decision logic for cycle selection.
Related Article: What is Moist Heat Sterilization?
Why a Structured Decision Tree for Heat Moist Sterilization Is Necessary
Despite the clarity of modern regulatory requirements, the application of moist heat sterilization in pharmaceutical manufacturing often remains inconsistent and overly simplified.
Regulatory guidance defines “what” must be achieved, but it does not provide a practical mechanism for making consistent, defensible decisions across different load types, container systems, and sterilization technologies.
In practice, manufacturers are routinely faced with critical questions such as:
- When is a gravity displacement cycle scientifically justified?
- When does a load require a pre-vacuum porous load cycle?
- Under what conditions is F₀ an appropriate lethality metric, and when is it misleading?
- When should steam–air mixtures or super-heated hot water shower processes be selected instead of saturated steam?
- How should validation and routine batch release criteria differ between solid loads and liquid products?
Without a structured decision logic, these questions are often answered based on historical practice, equipment availability, or perceived regulatory conservatism, rather than on sterilization science and risk assessment. This can result in:
- Inappropriate cycle selection
- Misalignment between the validation strategy and the routine release criteria
- Over-reliance on calculated lethality where physical limitations dominate
- Increased inspection findings and prolonged regulatory justification
A particularly common failure mode is the misapplication of F₀ as a universal acceptance criterion. Where air removal and steam penetration are the dominant risks (e.g., gravity and porous load cycles), F₀ calculations may give a false sense of assurance if steam penetration has not been conclusively demonstrated.
| Practical problem (what happens in real GMP manufacturing) | Typical “uninformed” decision basis | What goes wrong (risk / failure mode) | Decision tree outcome (what it fixes) |
|---|---|---|---|
| Cycle selection varies by site/equipment rather than science | Historical practice, “what we’ve always used,” equipment availability | Inappropriate cycle selection (wrong cycle for load/container) | Cycle choice is tied to the dominant mechanism (penetration vs heat transfer vs pressure-controlled integrity) |
| Validation evidence doesn’t match the true mechanism | Generic validation template applied to all cycles | Validation becomes non-mechanistic and hard to defend | Validation strategy is cycle-specific and mechanism-appropriate |
| Routine release criteria mirror validation activities | Conservative mindset; “repeat what we validated” | Misalignment between validation and routine release; unnecessary burden | Clear separation: validation proves capability; routine release confirms execution |
| F0 treated as universal acceptance criterion | Over-reliance on calculations and chamber data | False assurance where steam penetration is the real risk | F0 use is conditional: product-temperature based where appropriate; not primary for penetration-driven cycles |
| Solid loads and liquids handled with the same logic | “Steam is steam” simplification | Overprocessing or underprocessing; missed cold spots | Decision logic differentiates solids vs liquids and links to correct evidence |
| Overpressure / container-protective needs ignored | Focus only on temperature attainment | Container deformation, closure risk, damage | Triggers selection of SAM or hot water shower where pressure control is critical |
| Difficult regulatory justification during inspections | Post-hoc rationales; fragmented documents | Findings, prolonged responses, inconsistent narratives | An auditable, repeatable rationale linking cycle selection → validation → routine release |
Conversely, for liquid and container-protective processes, failure to use product-temperature–based lethality metrics may lead to unnecessary overprocessing or container damage.
Regulatory frameworks such as EU GMP Annex 1, ISO 17665-1, and the EMA sterilisation guideline implicitly require manufacturers to align the selection of sterilization processes, validation evidence, and routine release criteria with the dominant sterilization mechanism. However, this alignment is rarely made explicit in GMP documentation.
To address this gap, a Moist Heat Sterilization Process Decision Tree was developed to provide a clear, systematic, and auditable logic for:
- Selecting the appropriate moist heat sterilization process
- Defining the correct validation approach
- Establishing scientifically justified routine batch release criteria
This decision logic transforms regulatory expectations into repeatable GMP practice and forms the foundation for the framework presented in the next section.
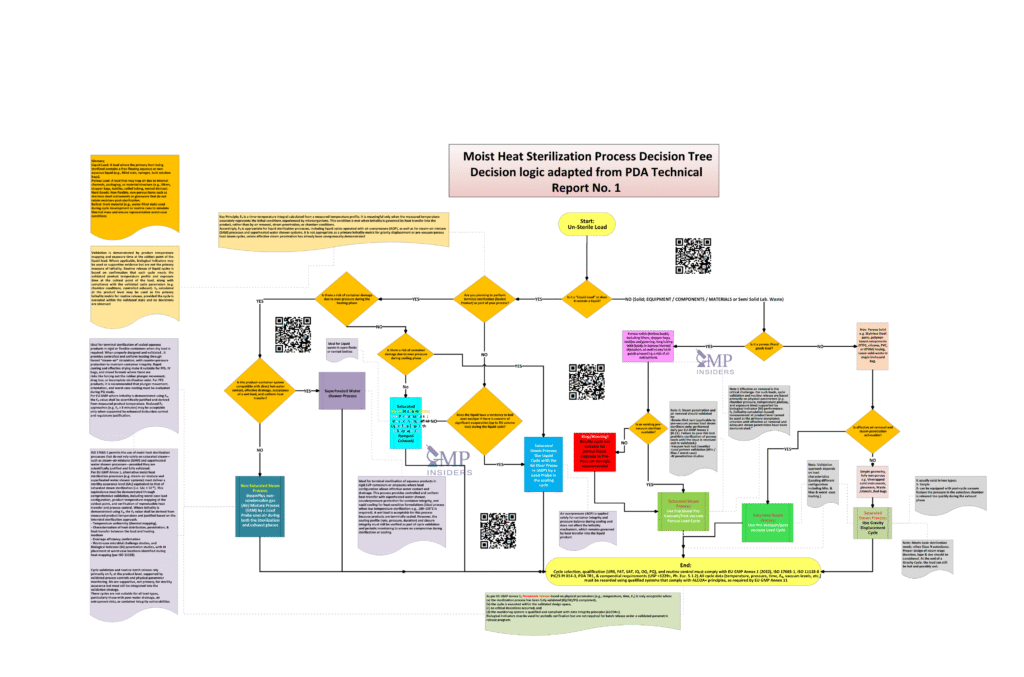
Risk-Based Decision Tree for Moist Heat Sterilization
To translate regulatory expectations and sterilization science into practical GMP execution, a Moist Heat Sterilization Process Decision Tree was developed. The decision tree provides a structured, risk-based logic to support selecting the appropriate moist heat sterilization process and to define the corresponding validation and routine batch-release strategy.
The decision tree is not intended to replace regulatory guidance, validation protocols, or scientific judgment. Rather, it serves as a decision-support framework that ensures critical regulatory and scientific considerations are consistently and transparently addressed.
*For a detailed view of the flowchart, click the image to open it in a new window.
Purpose of the Decision Tree
The primary objectives of the decision tree are to:
- Ensure load characteristics (solid, porous, liquid, sealed container) are evaluated before cycle selection
- Identify the dominant sterilization risk (air removal, heat transfer, or container integrity)
- Guide the selection of the appropriate moist heat process
- Align cycle validation and routine batch release criteria with the underlying sterilization mechanism
- Prevent inappropriate or unjustified use of lethality metrics, such as F₀, forcing these questions to be answered in a defined sequence, the decision tree reduces reliance on historical practice and improves regulatory defensibility.
Scope and Applicability
The decision tree applies to moist heat sterilization processes used for pharmaceutical and bio-pharmaceutical products, including:
- Gravity displacement steam cycles
- Pre-vacuum porous load steam cycles
- Saturated steam liquid cycles
- Steam–air mixture (SAM) process
- Super-heated hot water shower process
There is a clear separation between the following processes;
- Air-removal–limited processes (gravity, pre-vacuum & fraction pre-vacuum as well as post-vacuum approach)
- Heat-transfer–limited processes (liquid cycles),
- Container-protective processes (AOP, SAM, Superheated Water Shower)
It is applicable during:
- Process design and cycle selection
- Sterilization validation and re-qualification
- Change management (e.g. new container, load configuration, or sterilizer)
- Regulatory inspection preparation and documentation
Placement and Use of the Decision Tree
The decision tree should be positioned in GMP documentation after the regulatory justification section and before detailed validation requirements. Typical applications include:
- Validation Master Plans (VMP)
- Sterilization strategy documents
- Contamination Control Strategy (CCS)
- Sterilization SOPs and technical justification reports
What the Decision Tree Does Not Do
For clarity, the decision tree does not:
- Define specific cycle parameters (temperature, time, pressure)
- Replace load-specific validation studies
- Override product-specific risk assessments
Instead, it ensures that the correct questions are asked first, so that subsequent validation activities are technically appropriate and regulator-aligned.
Decision Tree Logic: Load Classification and Risk Identification
The foundation of the Moist Heat Sterilization Process Decision Tree is the principle that sterilization effectiveness is governed by the dominant physical mechanism acting on the load. Accurately identifying this mechanism is essential before selecting a sterilization cycle, defining validation requirements, or establishing routine batch-release criteria.
Accordingly, the first decision point in the tree is load classification, followed by identification of the dominant sterilization risk.
Load Classification
The decision tree requires loads to be classified into one of the following categories:

- Solid / Hard Goods (Non-porous): Examples include unwrapped metal instruments, simple glassware, and items without air-retaining geometry.
- Porous or Hollow Loads: Examples include textiles, filters, tubing, stopper bags, hoses, and complex hard goods where air entrapment is likely due to porosity, narrow internal diameters, bends, or layered structures.
- Heat-transfer–limited processes/Liquid Loads: (Open or Vented Containers) Liquids sterilized in containers that allow pressure equalization with the chamber, where heat transfer into the liquid governs lethality.
- Container-protective processes (Sealed Liquid Products): Liquids in sealed rigid or flexible containers (e.g. vials, BFS containers, PFS) where internal pressure, container integrity, and heat transfer must be controlled simultaneously using SAM or Super-heated hot water shower sterilizers
This classification aligns with EU GMP Annex 1 requirements that sterilization processes be appropriate for the load and container system (Annex 1 §8.57–§8.62).
Identification of the Dominant Sterilization Risk
Once the load is classified, the decision tree explicitly identifies the dominant sterilization risk, which determines the appropriate process and acceptance criteria:

a) Air Removal and Steam Penetration
For gravity displacement and pre-vacuum porous load cycles, incomplete air removal represents the primary risk. Residual air can prevent contact between saturated steam and the load, resulting in cold spots and ineffective sterilization.
Where air removal is the dominant risk:
- Demonstration of steam penetration is mandatory
- Physical parameters and BI performance are primary validation tools
- F₀ cannot be used as a primary acceptance criterion until steam penetration is proven
This reflects Annex 1 requirements for air removal verification and porous load control ( §8.57–§8.58)
b) Heat Transfer into the Product
For liquid cycles and container-protective processes, lethality is governed by heat transfer into the product, not chamber temperature.
Where heat transfer is the dominant risk:
- Product temperature profiles must be established at the coldest point of the load
- F₀ is calculated from measured product temperatures, not chamber conditions
- Validation focuses on heating, holding, and cooling behaviour
This approach is explicitly supported by Annex 1 for fluid sterilization (Annex 1 §8.59).
c) Container Integrity Under Pressure
For steam–air mixture and super-heated hot water shower processes, the dominant risk is loss of container integrity due to pressure differentials during heating and cooling.
Where container integrity is the dominant risk:
- Independent control of pressure and temperature is required
- Product temperature mapping must be combined with pressure control verification
- Lethality is demonstrated using product-based F₀, supported by heat transfer studies
This reflects Annex 1 expectations for non-rigid or heat-sensitive containers and for alternative moist heat systems (§8.62, §8.64–§8.65).
Why This Step is Critical
Failure to correctly identify the dominant sterilization risk often leads to:
- Misuse of F₀, where air removal is unproven
- Inappropriate cycle selection for the load
- Over-processing or container damage
- Weak or indefensible validation strategies
By explicitly separating load type from dominant risk, the decision tree ensures that cycle selection, validation, and routine release criteria are scientifically aligned.
Not All Moist Heat Cycles Are Created Equal
EU GMP Annex 1 clearly recognizes that moist heat sterilization is not limited to saturated steam processes. Section §8.55 explicitly permits the use of non-saturated moist heat processes, including:
- Steam–air mixture (SAM)
- Super-heated hot water, such as cascade or immersion Sterilizers
Section §8.55 states:
“Moist heat sterilization can be achieved using steam, but also includes other systems such as super-heated water systems, which may be used for containers that could be damaged by other cycle designs.”
However, this regulatory flexibility comes with a critical condition: these alternative processes must deliver a Sterility Assurance Level (SAL) ≤ 10⁻⁶ equivalent to saturated steam sterilization.
As a result, Sections §§8.64–8.65 require enhanced validation for such processes, including, as applicable:
- Full load temperature mapping
- Heat penetration and reproducibility studies
- Demonstration of uniform heating across the entire load
- Verification of process control under worst-case loading conditions
For porous and hard-goods loads, Annex 1 maintains a fundamentally different expectation. Section §8.57 identifies time, temperature, and pressure as the critical control parameters, while §8.61 requires routine verification of air removal and steam penetration.
Specifically, §8.61 states:
“There should be adequate assurance of air removal… For autoclaves, this should include an air removal test cycle (normally performed on a daily basis)…”
This requirement is typically met using recognized air-removal and steam-penetration tests, such as the Bowie–Dick test, DART, Lantor Cube, or Browne’s test, as described in PDA TR-1 and TR-48.
For heat-sensitive or non-rigid containers (e.g. BFS, FFS, flexible plastic containers), Section §8.62 requires:
“…appropriate cycle design and control (for instance, setting correct pressure, heating and cooling rates and loading patterns).”
This reinforces that container integrity, not air removal, may be the dominant risk for these applications.
Key Regulatory Implication
While Annex 1 permits multiple moist heat sterilization technologies, these cycles are not interchangeable. Each process is suitable only for specific load types and risk profiles.
In particular:
- Steam–air mixture and super-heated hot water processes are not suitable for loads with poor drainage or significant air-entrapment risk
- Porous and hollow loads require demonstrated air removal and steam penetration
- Liquid and container-protective processes require product-level heat transfer control
This distinction underpins the need for a structured decision logic, as implemented in the Moist Heat Sterilization Process Decision Tree.
Cycle Selection Across Moist Heat Sterilization Processes
Once the load type and dominant sterilization risk are identified, the decision tree guides selection of the most appropriate moist-heat sterilization process. This step is critical, as the effectiveness of any subsequent validation or routine release strategy depends on selecting a cycle that can physically control the dominant risk.
The decision tree distinguishes five primary moist-heat sterilization processes, each with a defined scope of application.
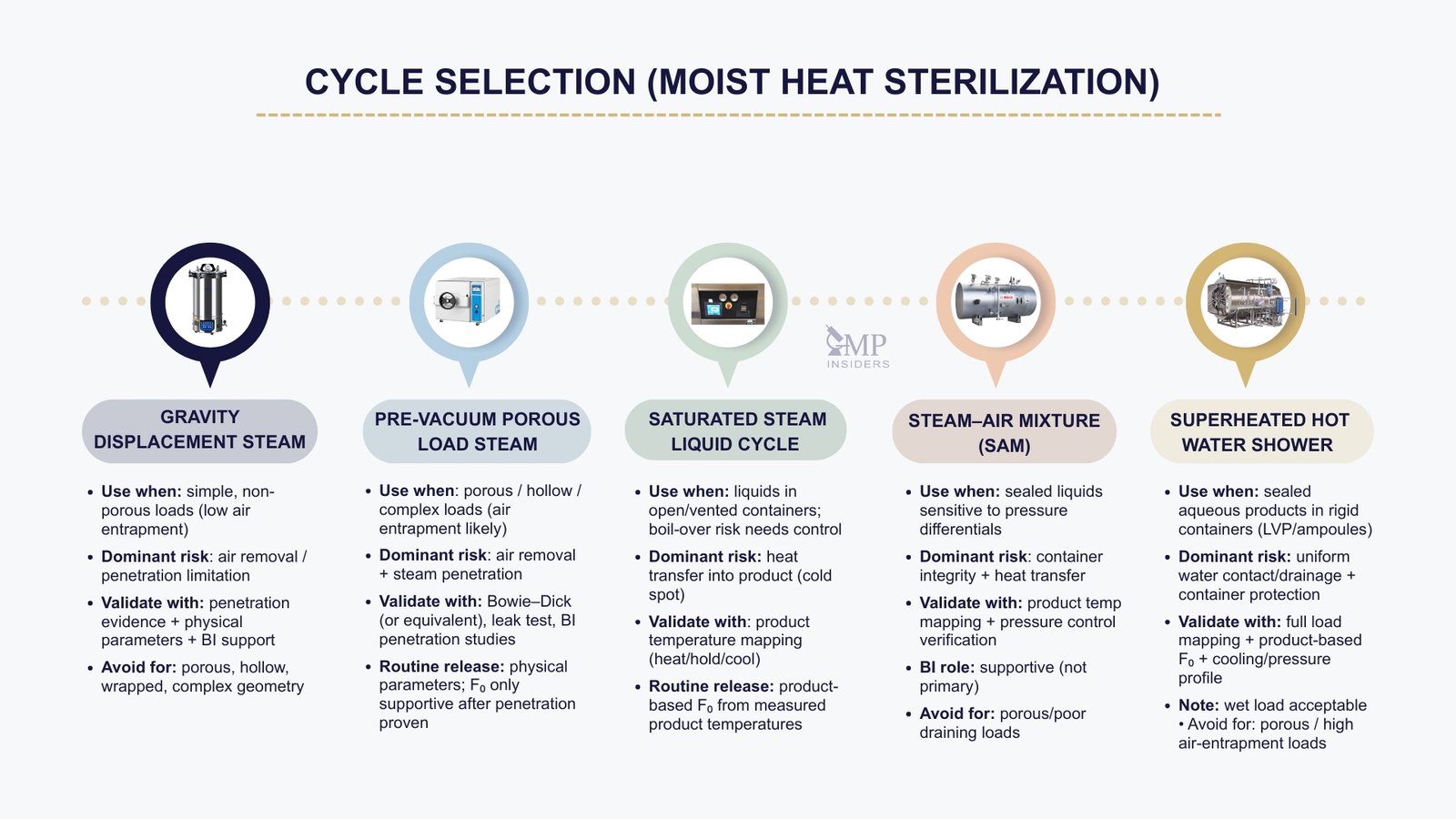
Gravity Displacement Steam Cycles
Gravity displacement cycles rely on passive displacement of air by incoming steam and are therefore limited in their ability to remove residual air. As a result, their use is restricted to simple, non-porous loads with minimal risk of air entrapment.
Appropriate applications include:
- Unwrapped metal instruments
- Simple glassware
- Loads without complex geometry or internal voids
For gravity cycles:
- Air removal is the dominant risk
- Validation must demonstrate effective steam penetration
- Physical parameters (time, temperature, pressure) supported by biological indicators form the primary validation basis
- F₀ cannot be used as a primary acceptance criterion unless steam penetration has been conclusively demonstrated
Gravity displacement cycles are not suitable for porous, hollow, or wrapped loads.
Pre-Vacuum Porous Load Steam Cycles
Pre-vacuum cycles are designed to actively remove air prior to steam exposure and are therefore appropriate for porous and hollow loads where air entrapment is likely.
Typical applications include:
- Textiles and gowning
- Cartridge filters and rubber stopper (bung) bags
- Tubing, hoses, and complex hard goods
For pre-vacuum cycles:
- Air removal and steam penetration remain the dominant risks
- Validation requires Bowie–Dick or equivalent testing, vacuum leak testing, and BI penetration studies
- Routine release is based on physical parameters, with F₀ used only as a supportive metric once penetration has been established
These cycles represent the standard of choice for porous loads under Annex 1 expectations.
Saturated Steam Liquid Cycles
Liquid cycles are fundamentally different from porous load cycles. Here, lethality is governed by heat transfer into the liquid, not by steam penetration into a porous structure.
Appropriate applications include:
- Aqueous products in open or vented containers
- Liquids requiring controlled exhaust to prevent boil-over
For liquid cycles:
- Product temperature profiles must be measured at the coldest point of the load
- Validation demonstrates heating, holding, and cooling behaviour
- F₀ is calculated from the measured product temperature profile, not from chamber conditions
- Routine release is based on confirmation that the validated product temperature profile and F₀ are achieved
This approach aligns directly with Annex 1 §8.59.
Steam–Air Mixture (SAM) Process
The steam–air mixture process allows independent control of temperature and pressure, making them suitable for sealed liquid products and containers sensitive to pressure differentials.
Typical applications include:
- Pre-filled syringes (PFS)
- Blow fill seal (BFS) and Form fill seal (FFS) containers
- Flexible or semi-rigid sealed containers (IV bags)
For SAM process:
- Container integrity and heat transfer are the dominant risks
- Validation is based on product temperature mapping and F₀ calculation at the product level
- Pressure control during heating and cooling is a critical validation element
- BI use is supportive, not primary
SAM processes are not appropriate for porous or poorly draining loads.
Super-heated Hot Water Shower Process
Super-heated water shower process relies on direct contact with recirculating hot water to deliver uniform and efficient heat transfer to the product. These systems are typically applied to sealed aqueous products in rigid containers, where effective water contact and drainage can be assured.
This process is particularly suitable for the terminal sterilization of aqueous products in rigid large-volume parenteral (LVP) containers or ampoules, provided that the load configuration allows uniform water distribution and complete drainage. Super-heated hot water shower processes offer several advantages, including controlled and homogeneous heat transfer, counter-pressure protection to maintain container integrity, and rapid, controlled cooling, making them well-suited for heat-sensitive formulations or applications requiring lower sterilization temperatures (e.g., 100–105 °C).
In contrast to saturated-steam processes, a wet load is acceptable for this process, as the products are terminally sealed prior to sterilization and are not adversely affected by direct water contact.
For the super-heated hot water shower process:
- Uniform water distribution and effective drainage must be demonstrated across the full load.
- Validation includes full load temperature mapping and F₀ calculation based on measured product temperatures at the coldest locations.
- The cooling profile (rate, pressure control and duration), as well as container closure integrity, must be verified as part of cycle validation.
- Biological indicators are used in a confirmatory role (e.g. during validation and periodic monitoring) to ensure no loss of sterility assurance during heating or cooling, but are not the primary basis for routine batch release.
As with steam–air mixture or super-heated hot water shower processes are not suitable for porous loads or loads with a high risk of air entrapment, where effective air removal and steam penetration are required.
Key Decision Tree Principle
A central principle reinforced by the decision tree is that:
No moist heat sterilization cycle is universally applicable. Each process is effective only when applied to loads whose dominant risks it can physically control.
By explicitly linking cycle selection to load type and dominant risk, the decision tree prevents inappropriate cycle application and provides a sound foundation for validation and routine batch release, as addressed in the next section.
Validation Strategy by Cycle Type
Validation of moist heat sterilization processes must be mechanism-appropriate. Applying a single validation model across all steam-based processes is neither scientifically justified nor aligned with EU GMP Annex 1, ISO 17665-1, or PDA guidance.
The Moist Heat Sterilization Process Decision Tree explicitly links each cycle type to a validation strategy that reflects the dominant sterilization mechanism: air removal and steam penetration, heat transfer into the product, or container integrity under pressure.
| Cycle Type | Dominant Mechanism / Main Risk | Validation Focus (What Must Be Proven) | Primary Evidence & Tests | BI Role | Routine Control |
|---|---|---|---|---|---|
| Gravity Displacement Steam | Passive air removal → residual air may block steam penetration | Demonstrate effective steam penetration at worst-case locations; verify stable time/temperature/pressure | Physical parameter verification; BIs at worst-case air traps; correlation of BI results with physical data | Primary during validation; not used for routine release | Control of validated physical parameters and load configuration |
| Pre-Vacuum Porous Load Steam | Active air removal → failure prevents penetration into porous/hollow loads | Confirm vacuum integrity and air removal; demonstrate penetration and heat transfer into hardest-to-sterilize locations | Vacuum leak test; Bowie–Dick (or equivalent); heat penetration mapping; BIs at worst-case locations; reproducibility across runs | Essential during validation; not part of routine release | Routine monitoring of air removal performance and physical parameters |
| Saturated Steam Liquid Cycles | Heat transfer into liquid → cold spot governs lethality | Map product cold spot; characterize heating, holding, and cooling; prove lethality via product temperature | Product probes; load mapping; F0 calculated from product temperature profiles | Supportive/confirmatory only; not primary evidence | Routine control via validated cycle parameters and consistent product/load configuration |
| Steam–Air Mixture (SAM) | Independent pressure and temperature control needed → container integrity risk | Demonstrate product temperature lethality while maintaining container integrity and proper P/T control | Product mapping; product-based F0; verification of pressure–temperature control; container closure integrity testing | Confirmatory during validation and periodic checks; not primary acceptance criterion | Routine control via P/T monitoring and periodic container integrity verification |
| Super-Heated Hot Water Shower | Heating via water distribution → risk of non-uniform temperature | Demonstrate uniform heat distribution, product lethality, and closure integrity under full load | Full-load temperature mapping; product-based F0; verification of water distribution, drainage, and cooling profiles; closure integrity tests | Confirmatory during validation and periodic checks; not used for routine release | Routine control via water system performance and validated temperature distribution |
Gravity Displacement Steam Cycles
Gravity displacement cycles rely on passive air removal and therefore have inherent limitations in removing residual air.
Validation focus;
- Verification of time, temperature, and pressure
- Placement of biological indicators (BIs) at worst-case locations to demonstrate steam penetration
- Correlation of BI performance with physical parameters
Role of BIs;
- Primary tool during validation to demonstrate process capability
- Used to confirm effective steam penetration
- Not used for routine batch release
F₀ calculations are not appropriate as a primary validation criterion unless effective steam penetration has been conclusively demonstrated.
Pre-Vacuum Porous Load Steam Cycles
Pre-vacuum cycles are designed to actively remove air and enable steam penetration into porous and hollow loads.
Validation focus;
- Vacuum leak testing and confirmation of vacuum performance
- Air removal testing (e.g. Bowie–Dick or equivalent)
- Worst case assessment & Heat penetration study
- Average probes temperature and BI studies demonstrating proper and effective heat penetration into the most difficult-to-sterilize locations/ items
- Confirmation of stable and reproducible physical parameters
Role of BIs;
- Essential during validation to demonstrate steam penetration
- Used to establish process capability
- Not part of routine batch release
Physical parameters remain the primary routine control once penetration has been validated.
Saturated Steam Liquid Cycles
For liquid cycles, lethality is governed by heat transfer into the liquid, not by air removal.
Validation focus
- Product temperature mapping at the coldest point of the load
- Evaluation of heating, holding, and cooling profiles
- Calculation of F₀ from measured product temperature profiles
Role of BIs;
- Supportive or confirmatory during validation
- May be used to provide additional assurance
- Not the primary demonstration of lethality
Here, product temperature measurement provides the most representative evidence of sterilization effectiveness.
Steam–Air Mixture (SAM) Process
SAM process requires independent control of temperature and pressure to protect container integrity while achieving sterilization.
Validation focus;
- Product temperature mapping under worst-case loading
- Verification of pressure–temperature control during heating and cooling
- Product-level F₀ calculation
- Container closure integrity verification
Role of BIs
- Confirmatory during validation and periodic monitoring
- Used to ensure no compromise during heating or cooling
- Not a primary acceptance criterion
Super-heated Hot Water Shower Process
Super-heated hot water shower systems rely on direct water contact to achieve uniform heating of sealed products.
Validation focus
- Full load temperature mapping to confirm uniform heat distribution
- Product-based F₀ calculation
- Verification of water distribution, drainage, and cooling profiles
- Confirmation of container closure integrity
Role of BIs;
- Confirmatory, supporting assurance of process robustness
- Used during validation and periodic checks
- Not used for routine batch release
Fundamental Biological Indicators Principle
Across all moist heat sterilization processes:
Biological indicators are tools to demonstrate process capability during validation. They do not replace physical measurements and are not used as routine batch release criteria.
This distinction is fundamental to compliance with EU GMP Annex 1, ISO 17665-1, and PDA TR-1 / TR-48.
Routine Batch Release Strategy
Routine batch release for moist heat sterilization processes must be based on process parameters that directly control the dominant sterilization mechanism. In line with EU GMP Annex 1, routine release criteria must be scientifically justified, consistently applied, and clearly differentiated from validation activities.
A key regulatory principle is that routine batch release confirms adherence to the validated process, rather than re-demonstrating sterilization capability.

General Principles
Across all moist heat sterilization processes:
- Routine release is based on verification of critical process parameters demonstrated during validation.
- Biological indicators are not used for routine batch release, as they do not provide real-time confirmation of cycle performance.
- Acceptance criteria must reflect the dominant sterilization mechanism for the selected cycle.
Routine Release by Cycle Type
The routine release strategy must be aligned with the dominant sterilization risks inherent to each cycle type, as these risks determine which parameters provide meaningful evidence of sterility assurance during routine operation.
Gravity Displacement and Pre-Vacuum Porous Load Cycles
For gravity displacement and pre-vacuum porous load steam cycles, the dominant risks are air removal and steam penetration.
Routine batch release is therefore based on:
- Verified time, temperature, and chamber pressure
- Confirmation of cycle reproducibility
- Review of critical cycle alarms and deviations
Once effective air removal and steam penetration have been validated, parametric release may be applied, subject to regulatory requirements.
Saturated Steam Liquid Cycles
For liquid cycles, lethality is governed by heat transfer into the product.
Routine batch release is based on:
- Confirmation that the validated product temperature profile has been achieved
- Verification of F₀ calculated from measured product temperatures
- Review of heating, holding, and cooling phases
Chamber temperature alone is insufficient for routine liquid-cycle release.
Steam–Air Mixture (SAM) Process
For SAM process, both heat transfer and pressure control are critical.
Routine batch release is based on:
- Verified product-level F₀ calculated from mapped thermocouple locations
- Confirmation of pressure control throughout heating and cooling
- Review of container integrity-related alarms
Super-heated Hot Water Shower Process
For super-heated hot water shower process, routine release focuses on uniform heating and container protection.
Routine batch release is based on:
- Verified product-level F₀ derived from validated thermocouple locations
- Confirmation of water temperature control, circulation, and cooling profile
- Review of container integrity indicators
What Routine Batch Release Does NOT Include
To avoid misinterpretation, routine batch release does not include:
- Routine use of biological indicators
- Repetition of the validation-phase thermal mapping
- Re-establishment of worst-case conditions
These activities fall under validation or requalification, not routine release.
Summary Table: Validation and Routine Batch Release Across Moist Heat Sterilization Processes
One of the recurring challenges in pharmaceutical sterilization is the inconsistent interpretation of validation and routine batch-release requirements across different moist-heat processes. Although regulatory guidance clearly distinguishes between sterilization mechanisms, these distinctions are often lost when processes are grouped generically as “steam sterilization.”
To address this gap, Table 1 provides a side-by-side comparison of the major moist heat sterilization cycles discussed in this article. The table consolidates, in a single view, the dominant process risk, the primary basis for validation, the role of biological indicators, and the routine batch release criteria for each cycle type.
Summary Table – Cycle Validation & Routine Release (All Moist-Heat Cycles)
| Moist Heat Sterilization Process | Typical Load / Application | Dominant Process Risk | Primary Basis for Validation | Role of Biological Indicators (BI) | Routine Batch Release Basis |
|---|---|---|---|---|---|
| Gravity Displacement Steam Cycle | Simple non-porous solids (e.g., unwrapped instruments, simple glassware) | Passive air removal | Physical parameters (time, temperature, pressure) supported by steam penetration studies | Primary during validation to demonstrate penetration at worst-case locations; not used for routine release | Verified physical parameters (parametric release where permitted) |
| Pre-Vacuum Porous Load Steam Cycle | Porous or complex loads (textiles, filters, tubing, stopper bags) | Air removal and steam penetration | Physical parameters supported by air removal testing and penetration studies | Primary during validation to demonstrate effective penetration; not used for routine release | Verified physical parameters (time, temperature, pressure) |
| Saturated Steam Liquid Cycle (Open or Vented Containers) | Aqueous liquids in open or vented containers | Heat transfer into liquid | Product temperature profile at coldest point and F0 calculated from measured product temperatures | Supportive/confirmatory during validation only; not primary proof | Verified product temperature profile and F0 derived from product temperature |
| Saturated Steam Liquid Cycle (Sealed Containers) | Sealed liquid products tolerant of saturated steam | Heat transfer and container stress | Product temperature mapping and F0 from measured product temperatures | Supportive/confirmatory during validation; not routine criterion | Verified product-level F0 and validated temperature profile |
| Steam–Air Mixture (SAM) Process | Sealed products requiring pressure control (e.g., PFS, BFS, flexible containers) | Heat transfer and container integrity under pressure | Product mapping, pressure–temperature control verification, and F0 from product temperatures | Confirmatory during validation and periodic monitoring; not routine criterion | Verified product-level F0 and pressure control within validated limits |
| Super-Heated Hot Water Shower Process | Sealed aqueous products in rigid containers (e.g., LVPs, ampoules) | Uniform heat transfer and container integrity | Full-load mapping, F0 from product temperatures, and verification of water distribution and cooling profile | Confirmatory during validation and periodic monitoring; not routine criterion | Verified product-level F0 and validated heating/cooling parameters |
Key Messages Reinforced by the Table
Several fundamental principles become immediately clear when the processes are viewed side-by-side:
- Not all steam-based processes are equivalent, and they cannot share the same validation or release logic.
- Biological indicators demonstrate process capability during validation but are not routine batch-release criteria.
- F₀ is not a universal metric. It is appropriate only for processes governed by product heat transfer (e.g. liquid, steam–air mixture, and super-heated hot water shower processes), and not for porous load cycles dominated by air removal.
- Physical parameters dominate porous load processes, while product-level measurements dominate liquid and container-protective processes.
These distinctions directly align with the expectations of EU GMP Annex 1, ISO 17665-1, and EMA sterilization guidance and serve as the technical backbone of the Moist Heat Sterilization Process Decision Tree.
The Rise of Parametric Release: Data Over Assumptions
Parametric release represents a significant shift in regulatory thinking, from reliance on endpoint testing or surrogate indicators to direct control and verification of critical process parameters. For moist heat sterilization processes that are well understood, robustly validated, and tightly controlled, parametric release is no longer an exception but an accepted and encouraged approach.
Regulatory Expectations for Parametric Release
EU GMP Annex 1 explicitly supports the use of parametric release for moist heat sterilization, particularly for fluid cycles.
Section §8.59 states:
“Validation of fluid cycles should include temperature, time and/or F₀. Critical processing parameters should be subject to defined limits and be confirmed as part of the routine cycle acceptance criteria.”
This makes it clear that, where lethality is demonstrated by measured product temperature and Fo, routine batch release may be based on process parameters rather than on biological indicators or sterility testing.
However, Annex 1 also emphasizes that parametric release is not automatic. Section §10.4 introduces an important additional expectation:
“For products authorised for parametric release, a supporting pre-sterilisation bioburden monitoring programme for the filled product prior to initiating the sterilisation cycle should be developed, and the bioburden assay should be performed for each batch.”
Together, these clauses establish that parametric release is acceptable only when supported by strong upstream and in-process controls.
Prerequisites for a Defensible Parametric Release Program
A robust parametric release program for moist heat sterilization requires, at a minimum:

- Successful IQ, OQ, and PQ, including worst-case load configurations and comprehensive temperature mapping
- Demonstrated F₀ at the coldest point of the product, based on measured product temperature (not chamber conditions)
- Robust control of critical process parameters, including pressure, heating and cooling ramp rates, and exhaust profiles
- No critical deviations during the sterilization cycle
- ALCOA+ compliant data systems, ensuring data integrity, traceability, and real-time review
- A defined and routine pre-sterilisation bioburden monitoring programme, where required by the product authorisation
Within a validated parametric release framework, biological indicators may be used for periodic or confirmatory verification, but they are not required for routine batch release, provided all acceptance criteria are met.
This approach reflects a transition from assumed sterility to demonstrated, data-driven sterility assurance.
Regulatory & Global Standard Expectations
The risk-based approach to moist heat sterilization is not an internal best practice but a direct consequence of converging regulatory and international standard expectations that collectively require manufacturers to align sterilization strategy, validation, and routine release with the actual sterilization mechanism and dominant risks.
EU GMP Annex 1 (2022)
Annex 1 explicitly broadens the scope of moist heat sterilization beyond saturated steam. It states that sterilization may be achieved using steam and other processes, such as a superheated hot-water shower, particularly for containers that may be damaged by conventional cycle designs (Annex 1, §8.62).
Key regulatory expectations relevant to moist heat sterilization include:
- Effective air removal and steam penetration must be ensured for porous and hard-goods loads, with routine verification and monitoring (Sections § § 8.57& 8.58).
- Time, temperature, and pressure are the primary controlling parameters for porous load sterilization processes (Section, §8.59).
- For fluid sterilization, Annex 1 explicitly allows the use of temperature, time and/or F₀ as acceptance criteria, recognizing the different heat transfer mechanisms involved (Section, §8.59).
- Non-rigid or heat-sensitive containers must be protected from deformation or damage by appropriate cycle design, pressure control, and controlled heating and cooling rates Section, §8.62).
- Super-heated hot water process require full-load temperature mapping and demonstration of uniform heat transfer and reproducibility (, §8.64 & §8.65).
These requirements make it clear that not all steam-based processes are equivalent, and that manufacturers are expected to justify why a specific moist heat process is selected for a given load and dominant risks, rather than applying a single default cycle.
EMA Guideline on Sterilization
The EMA Guideline on the Sterilization of the Medicinal Product, Active Substance, Excipient and Primary Container further refines expectations for steam sterilization by defining acceptable F₀ ranges, bioburden assumptions, and validation and dossier requirements for terminal sterilization and post-aseptic heat treatment.
The guideline clearly differentiates:
- Reference and overkill terminal steam sterilization cycles
- Reduced lethality cycles (F₀ ≥ 8 min) with increased validation burden
- Post-aseptic terminal heat treatment (F₀ < 8 min), which is not equivalent to terminal sterilization and requires full bioburden characterisation and justification
These distinctions are critical to understanding why steam sterilization cannot be treated as a single method and why different validation and routine release strategies are required.
See Also: Terminal Sterilization vs Aseptic Processing
FDA Perspective
While FDA guidance does not prescribe specific cycle designs, inspection experience consistently shows expectations for:
- Clear justification of cycle selection
- It is important to remove air from the autoclave chamber as part of a steam sterilization cycle.
- Demonstrated understanding of air removal limitations
- Mapping studies should be conducted with calibrated measurement devices i.e thermocouple.
- Alignment between validation evidence and routine release criteria
- Avoidance of unjustified reliance on calculated lethality where physical phenomena dominate
ISO 17665
ISO 17665 establishes the framework for the development, validation, and routine control of moist heat sterilization processes, including those that do not rely on saturated steam. It requires that:
- Sterilization processes should be selected based on product and load characteristics ( Clause 6)
- Validation and routine control reflect the actual sterilization mechanism, whether air removal, steam penetration, or heat transfer (Clauses 8–10)
- Lethality metrics such as F₀ (F sub zero) should be applied only where meaningful product temperature measurement is possible
This reinforces the expectation in Annex 1 for risk-based, load-specific decision-making.
FAQ
How Do I Justify Using a “Mixed Load” (Different Items in One Cycle) Without Creating Validation Risk?
Mixed loads are acceptable only if the sterilization mechanism and dominant risks remain controlled for every item in the load. The key is to define clear rules for load composition, load arrangement, and maximum/minimum load sizes.
Validation must demonstrate worst-case heat penetration and/or steam penetration study as well as BIs verification across the combined configuration, not individual items tested separately. If items differ substantially (porous, liquid, and sealed), the mixed load is often difficult to justify and should be separated into distinct, validated load families. Any routine deviation from the validated configuration should trigger a documented risk assessment and deviation procedure.
What Is a Defensible Approach to Defining “Load Families” for Sterilization?
A load family groups items that behave similarly with respect to air removal, steam penetration, heat transfer, and drainage. A defensible strategy is to define families by geometry (hollow/porous), packaging type (wraps, pouches, trays), mass/thermal inertia, and container closure behavior.
Once defined, select a worst-case representative load within each family and validate to that. The rationale must be explicit and consistent, so inspectors can see why certain items are equivalent. If new items are introduced, they should be evaluated against the family definition before inclusion.
What Change-Control Events Should Automatically Trigger a Sterilization Impact Assessment?
Any change that can affect air removal, steam quality, heat transfer, pressure control, or measurement reliability should trigger an assessment. Examples include changes to packaging (including supplier), load configuration, container/closure design, sterilizer control software, steam supply, filters, drain system, and probe calibration approach. Maintenance that affects vacuum performance or pressure regulation is also high impact.
The impact assessment should explicitly state whether the validated worst-case is still valid. Where uncertainty remains, confirmatory studies should be planned rather than relying on argumentation alone.
How Should I Handle a Failed Bowie–Dick (Or Equivalent Air Removal) Test in a GМP Setting?
A failed equipment air removal test should be treated as a potential loss of process assurance for porous-load cycles. The immediate question is whether any loads were processed since the last acceptable test and, if so, what sterilization claims were made for those loads. Typically, you quarantine affected loads, conduct a documented investigation, identify the assignable cause, and implement a CAPA.
You also need a decision on whether retesting and requalification are required before resuming use. The response should be aligned with your risk classification and validated operational controls, not improvised on a case-by-case basis.
When Is It Appropriate to Use Chemical Indicators (CIs), and What Are Their Limitations?
Chemical indicators are useful for demonstrating exposure to specific conditions (e.g., steam contact, temperature thresholds) and for supporting load-handling controls. They are not a substitute for validation evidence or physical measurements, and they cannot quantify lethality in a manner that supports batch-release decisions.
Their primary value is operational: confirming correct load placement, segregation, and that a load has undergone a cycle. If used, the CI type and its intended meaning must be clear in your procedure. Misuse occurs when CIs are interpreted as proof of sterility rather than process exposure.
Final Thoughts
Modern regulatory expectations make it clear that moist-heat sterilization is no longer a one-cycle-fits-all process. It is a critical element of the Contamination Control Strategy (CCS) and must be designed, validated, and controlled using a risk-based, science-driven approach.
Regulators now expect manufacturers to demonstrate:
- Risk-based cycle selection (e.g. gravity displacement, pre-vacuum porous load, liquid cycles, steam–air mixture, super-heated hot water)
- A clear and defensible linkage between validation and routine monitoring
- Decisions driven by process understanding and data, rather than legacy practice
The era of “we’ve always done it this way” is over. True compliance isn’t about following a recipe, it’s about understanding the science, controlling the risks, and proving it with data.
If your sterilization program still defaults to “121°C for 15 min” without asking “What am I really sterilizing and what could go wrong?”… It’s time to rethink!