Modern distribution networks are complex: routes span multiple climatic zones, products are increasingly temperature-sensitive, and regulators expect objective evidence that medicinal products can withstand the environmental conditions to which they are exposed during transit.
In this context, transport validation is not simply about temperature monitoring; it is about demonstrating, through scientifically designed and documented studies, that the transportation process is controlled, predictable, and consistently protects product quality.
From a GMP and GDP perspective, transportation is an extension of the manufacturing and quality system. Once a product leaves a warehouse, the responsibility to protect its quality does not diminish.
Regulators, particularly under EU GDP, WHO guidance, and aligned global expectations, emphasise that companies must understand transport-related risks, design appropriate controls, qualify logistics solutions, and verify that they continue to perform in real-world conditions.
This article provides a structured, practice-oriented interpretation of transport validation within pharmaceutical GDP. It explains what transport validation means, clarifies when it is required, outlines how it should be designed and executed, and connects it to risk management, documentation, deviation handling, and ongoing verification.
What Does Transport Validation Mean
Transport validation is often misunderstood or reduced to temperature monitoring alone. In reality, it is a structured, evidence-based activity that demonstrates that pharmaceutical products can be transported under defined conditions without compromising their quality.
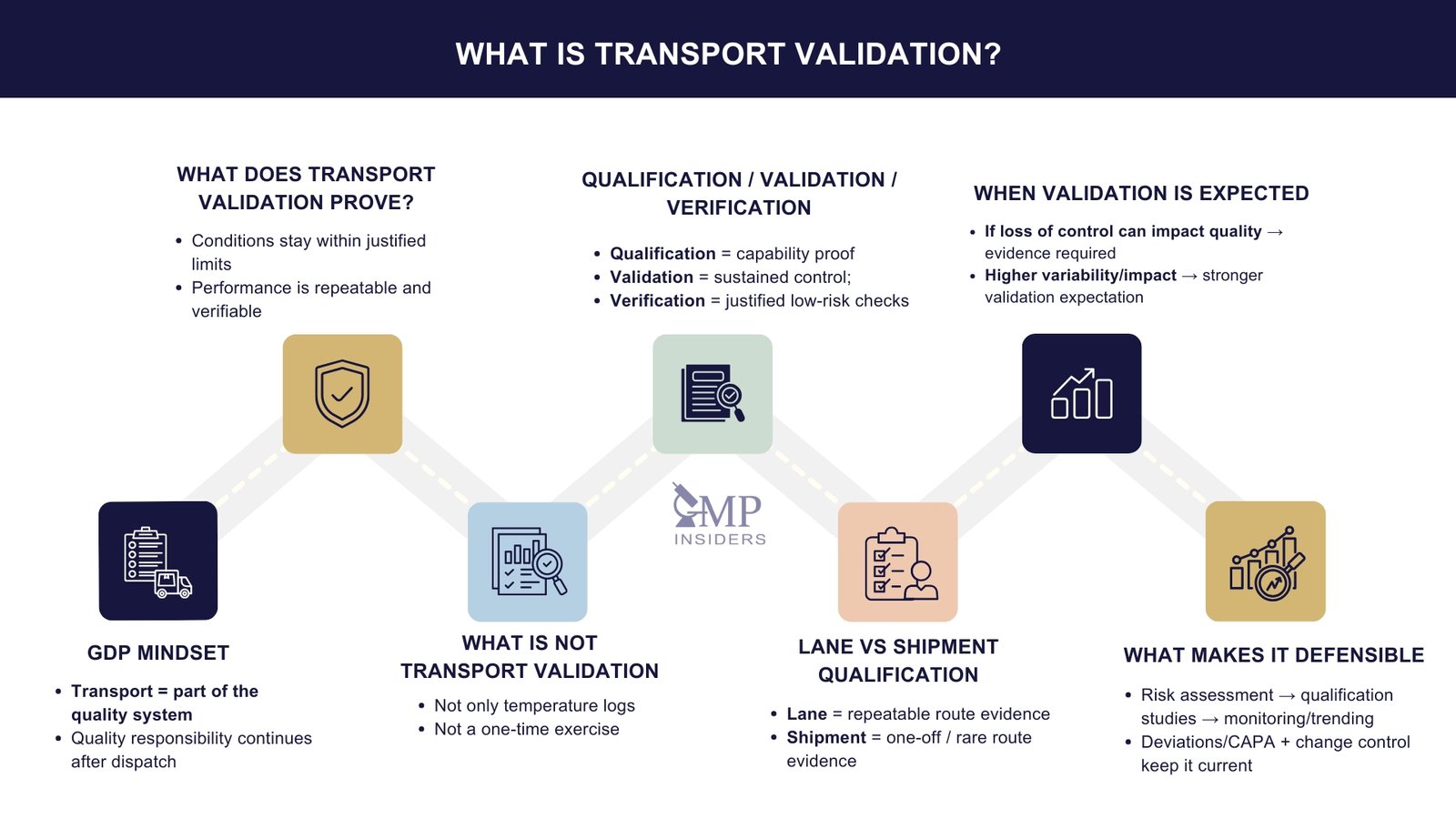
It confirms that transport routes, packaging solutions, processes, and controls are capable of consistently maintaining conditions aligned with product stability data and GDP requirements.
In practice, transport validation transforms transport from a “logistics event” into a controlled, qualified, and defensible quality process.
Definition in a GDP / GMP Context
Transport validation means providing documented, scientific evidence that:
- The transportation process can consistently maintain the required environmental conditions
- Products are protected from unacceptable risks during distribution
- Routes, packaging, vehicles, and handling processes are suitable for their intended use
- Controls are effective under normal and reasonably foreseeable stress conditions
- The outcome is predictable, repeatable, and verifiable
“Can we prove that our transport process protects our product every time, not just occasionally?”
What Is NOT Transport Validation
Misinterpretations are a common weakness seen in inspections. Transport validation is not:
- Not just temperature monitoring: Monitoring records alone do not prove capability; they only record what happened.
- Not limited to cold chain: Controlled room temperature products also require transport justification and, where needed, validation.
- It is not only about equipment; it is about the full system: routes, providers, conditions, packaging, handling, and governance.
- Not a one-time exercise: It follows lifecycle principles and requires periodic review and re-qualification.
- Not a “vendor says it works, so it’s fine” activity: Vendor qualification data can support, but the company remains responsible for assurance.
Qualification, Validation, and Verification in Transport
Terminology is frequently confused. A clear understanding helps define expectations and defend decisions during inspections.
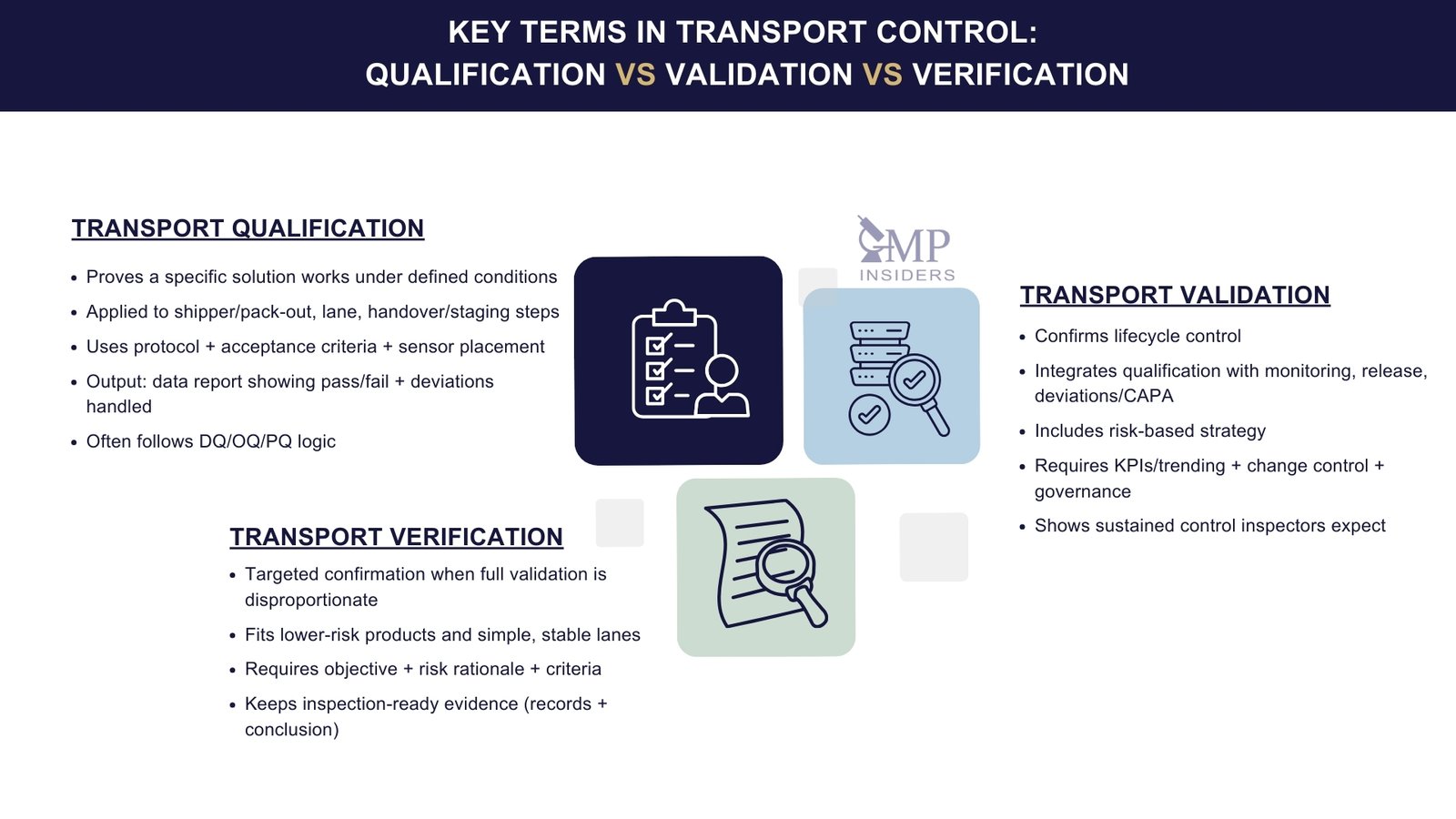
Transport Qualification
A transport qualification demonstrates that a specific transport solution performs as intended under defined, relevant conditions. It is “capability proof” for the technical and operational design you intend to use.
What qualification is typically applied to
- Packaging systems (shipper + coolant + pack-out configuration + loading pattern + closure method)
- A defined transport lane/route (or a defined distribution network segment)
- A transport process (handover steps, staging conditions, data retrieval, acceptance/release steps)
- A service provider’s ability to execute requirements (where provider performance is part of the control strategy)
What does qualification evidence usually look like in practice
- Design rationale: why this packaging and this lane strategy is selected (risk assumptions, boundary conditions, known constraints)
- Protocol-driven studies: predefined objective, study conditions, monitoring approach, placement strategy, acceptance criteria
- Data-backed reports: what happened, whether criteria were met, how deviations were handled, and what this means for routine use
How DQ/OQ/PQ thinking commonly fits
Even if you don’t formally label it, qualification often naturally follows DQ/OQ/PQ logic:
- DQ (Design Qualification): demonstrates that the proposed solution meets product requirements, is stable, is based on lane-exposure assumptions, and haspackaging capability. Deliverables typically include risk assessment, pack-out rationale, and defined acceptance criteria/boundary conditions.
- OQ (Operational Qualification): tests the solution in a controlled manner against defined challenges (simulated profiles, controlled holds, stress points) to show it performs within limits under expected/worst-case assumptions.
- PQ (Performance Qualification): confirms performance under real operational conditions, typically across multiple shipments to demonstrate repeatability and to reveal operational weak points (handover discipline, dwell times, handling variability).
Transport Validation
Transport validation is broader and more “system-level.” It provides lifecycle confirmation that transport processes consistently perform as intended and remain under control over time. It integrates qualification results, routine performance monitoring, deviation management, and governance into one defensible control story.
What validation covers beyond qualification
Qualification shows capability at a point in time; validation adds the elements that keep the process controlled after go-live:
- Risk-based strategy: documented rationale for how much evidence is needed for different products/lanes (and why)
- Process integration: how packaging, logistics steps, monitoring, release decisions, and deviation handling work together
- Ongoing control mechanisms: trend review, lane performance KPIs, periodic evaluation, change control triggers
- Quality governance: responsibilities, escalation paths, decision authority, supplier oversight, and documented review frequency
What “validated” should mean in a GDP/GMP sense
A validated transport approach shows that:
- controls are defined and implemented (procedures + training + governance),
- performance is demonstrated (qualification + routine data),
- deviations feed back into improvement (investigation + CAPA + effectiveness checks),
- changes are assessed for impact (change control + requalification triggers)
This is the distinction inspectors care about: validation is credible when it demonstrates sustained control, not only initial performance.
Transport Verification
Transport verification is a targeted confirmation activity used when a full validation package would be disproportionate to risk, but objective evidence is still needed to demonstrate control for the defined scope.
Verification is often misunderstood as “lighter = minimal.” In practice, verification remains acceptable only when it is risk-based, justified, and documented with clear criteria.
When verification is typically appropriate
- Lower-risk products with robust stability margins and low sensitivity to foreseeable transport stress
- Simple lanes (short duration, few handovers, high predictability, stable infrastructure)
- Well-understood environments with credible historical performance data that supports the assumption of control
- Low variability in execution, where the process is stable and routinely demonstrated through monitoring/trending
What verification should still include
- A defined objective: what you are confirming and why
- A documented risk assessment and rationale that explains why verification is sufficient
- Defined acceptance criteria (including how excursions are evaluated and what triggers escalation)
- Retained evidence that is reviewable during inspection (records, monitoring results, conclusions)
Lane Qualification vs Shipment Qualification
These are often conflated, but they address different questions. Understanding where each applies helps you design a proportionate program and avoid overclaiming.
Lane Qualification
Lane qualification evaluates a defined, repeatable route (or a defined distribution pathway) as a system. It is the foundation for routine distribution because it provides evidence that the lane can be relied upon repeatedly within defined boundaries.
What lane qualification typically considers
- Climatic and seasonal exposure: where the lane runs, seasonal extremes, and known stress periods
- Duration and dwell time patterns: not only nominal time, but worst-case credible holds (hubs, customs, weekends)
- Handover complexity: number of transfers, control points, and who owns the chain at each step
- Infrastructure reliability: variability between mature vs developing lanes, known weak points, historical deviations
- Repeatability: evidence across multiple runs that performance is not “accidental”
Shipment Qualification
Shipment qualification applies to a one-off or rare shipment when lane qualification is not established, not feasible, or not appropriate.
Where shipment qualification commonly fits
- Clinical supplies and investigational products
- Emergency deliveries or special access shipments
- Remote destinations, non-standard routing, or unusual handling constraints
- Low-frequency shipments where building lane history is not realistic
| Aspect | Lane Qualification | Shipment Qualification |
|---|---|---|
| Core question | “Is this route/pathway reliable for routine use?” | “Is this one shipment controlled enough for its specific conditions?” |
| Scope | Defined, repeatable lane (route + pathway as a system) | One-off or rare shipment (case-by-case) |
| Typical use | Foundation for routine distribution | Used when lane qualification is not established, feasible, or appropriate |
| What it considers | Climatic and seasonal exposure, worst-case dwell times, handover complexity, infrastructure reliability, repeatability across runs | Shipment-specific constraints: routing uncertainty, special handling, limited controls, unique risks |
| Evidence style | Multiple runs with defined boundaries and assumptions to show performance is not “accidental” | Targeted, shipment-level evidence (plan, monitoring, documented outcome) |
| Common scenarios | Established lanes, recurring customers or markets, predictable routing with defined control points | Clinical or IMP supplies, emergency or special access, remote or non-standard routes, low-frequency shipments |
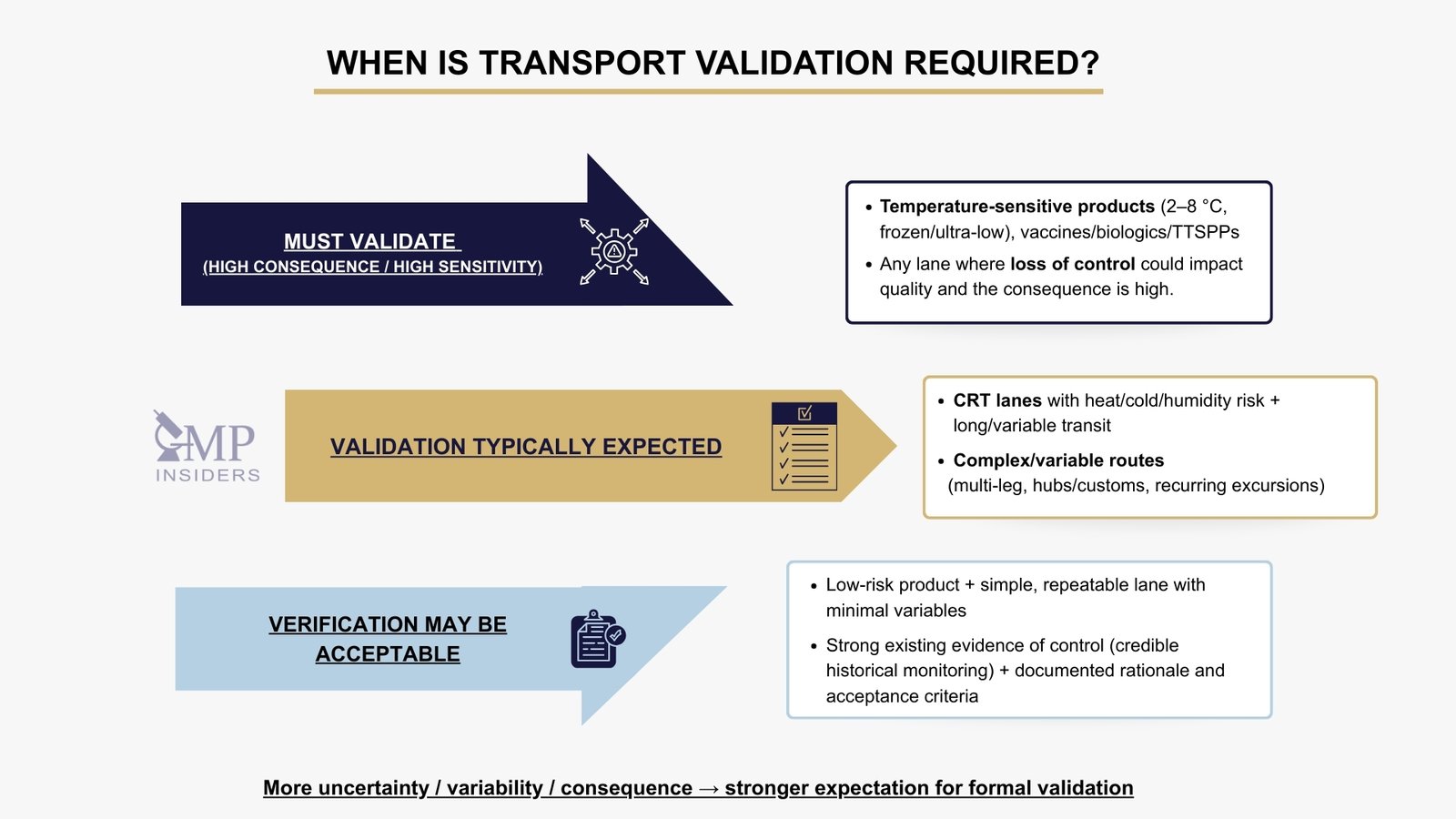
When Is Transport Validation Required?
Transport validation is not optional or discretionary; it is required whenever there is a credible risk that transportation conditions may impact product quality. The expectation is that companies use a risk-based, scientifically justified approach to determine where full validation, qualification, or verification is appropriate.
Regulators increasingly challenge companies that rely on assumptions or historical precedent rather than structured assessment and documented evidence.
Transport validation is required when transport conditions must be proven to remain suitable, when variability is expected, or when the consequences of failure are significant.
General GDP Expectation
Under GDP principles, transport must not expose medicinal products to conditions inconsistent with their stability profile or labeled storage requirements. Therefore, transport validation is required whenever:
- The transport environment may impact product quality
- There is uncertainty or variability in transport conditions
- The distribution system needs to demonstrate capability and control
- Regulatory expectations explicitly call for documented assurance
In other words, if transport conditions matter, they must be validated or justified.
Situations Where Transport Validation Is Typically Required
Transport validation is generally expected whenever distribution conditions could realistically influence product quality, safety, or stability.
In practice, the question inspectors ask is simple: could loss of control during transport have a meaningful impact on quality, and can you demonstrate that your controls are effective? If the answer is “yes,” then documented validation evidence (or a justified, risk-based alternative) is required.
Temperature-Sensitive Products
For products whose quality depends directly on maintaining defined temperature conditions, transport validation is a regulatory requirement. These medicines must have documented evidence that transportation controls are capable of consistently maintaining required storage conditions throughout the distribution journey.
Transport validation is unquestionably required for:
- Refrigerated products (2–8 °C)
- Frozen products (−20 °C, −70 °C, and ultra-low)
- Ultra-low temperature biologics
- Vaccines and other biological products
- Time- and temperature-sensitive pharmaceutical products (TTSPPs)
Why does this drive validation?
- These products can be highly sensitive to environmental change, sometimes with limited tolerance for excursions.
- Excursions may impact potency, impurity profile, physical stability, or functional performance, often with a direct link to patient risk.
- Regulators expect proactive assurance, not retrospective explanations. That means a defined strategy supported by qualification evidence, monitoring data, deviation handling, and governance that is scientifically justified and inspection-ready.
Controlled Room Temperature (CRT) / Ambient Products
A frequent misconception is that CRT products are automatically “low concern.” Many CRT products remain vulnerable to prolonged heat exposure, freezing events, high humidity, or repeated temperature cycling, especially when shipping patterns include long dwell times, summer peaks, or variable last-mile conditions.
Transport validation (or strong, documented justification for a lighter approach) is typically expected when:
- Transport environments regularly exceed the intended CRT range (e.g., repeated summer exposure in uncontrolled staging areas).
- The lane includes hot/humid regions, such as those classified as ICH Zone IV.
- Shipping duration is long or highly variable, increasing exposure time and uncertainty.
- Historical performance shows recurring deviations, excursions, or missing/weak monitoring data.
- Packaging relies on passive protection, and performance depends on proper pack-out and handling (processes that can drift without control).
High-Risk Distribution Scenarios
Even for products that appear stable, validation expectations increase sharply when distribution complexity or uncertainty increases. Complexity introduces more failure modes: more handovers, more uncontrolled dwell points, more variability, and less ability to intervene when conditions drift.
Transport validation is commonly expected for scenarios such as:
- Long-distance or international distribution, especially where customs and hub dwell times are significant.
- Multi-leg and multi-carrier supply chains, where chain-of-custody and consistent execution become harder to control.
- Routes crossing multiple climatic regions (where seasonal and geographic variability increase uncertainty).
- Transport involving high-variability modes or interfaces, including:
- Air freight (tarmac exposure, hub delays, unpredictable dwell time)
- Ocean freight (long durations, container variability, limited intervention options)
- Uncontrolled environments (staging areas, depots, third-party cross-docks)
- Distribution to remote or infrastructure-limited regions, where reliable cold chain capability and rapid escalation may not be guaranteed.
Rule of thumb: the greater the uncertainty and variability in the lane, the stronger the expectation for formal validation supported by robust qualification evidence, monitoring, deviation handling, and supplier governance.
When Verification May Be Acceptable Instead of Full Validation
A reduced approach may be justified when the transport activity is clearly low risk and there is reliable evidence that the required conditions are already maintained in a stable, controlled manner. Verification can be appropriate when:
- The product has low sensitivity to transport-related stress (temperature excursions, vibration, delays), and its quality attributes are not likely to be impacted under the expected conditions.
- The distribution route is short, uncomplicated, and repeatable, with minimal variables (same lane, same packaging configuration, same carrier, same handling steps).
- There is established evidence that conditions are under control, such as consistent monitoring data from previous shipments, qualification evidence from comparable lanes, or robust controls across the logistics chain.
- The decision is supported by a clear, documented rationale explaining why verification is proportionate to risk and why full validation would not provide meaningful additional assurance.
However, verification must still be treated as a controlled GMP/GDP activity:
- It does not mean “do nothing” or “skip controls.” It means using targeted checks that are appropriate for the risk profile.
- The verification strategy must be risk-based and defensible, with defined acceptance criteria, defined responsibilities, and a clear link to the underlying risk assessment.
- Evidence must still be generated, recorded, and retained in a way that is traceable and reviewable (what was checked, when, by whom, against which limits, and what actions are taken if results are not acceptable).
In practice, the credibility of verification depends on how well the organisation can demonstrate that the lane remains in control over time, not merely that it appeared acceptable at one point.
Risk-Based Approach to Transport Validation
Transport validation in the pharmaceutical industry is fundamentally risk-driven. Regulators expect companies to design their validation strategy based on an understanding of the risks associated with the product, distribution environment, and transport process rather than applying a uniform approach to every shipment.
A well-implemented risk-based approach ensures resources are focused where they matter most: on protecting patient safety and preventing compromise of product quality.
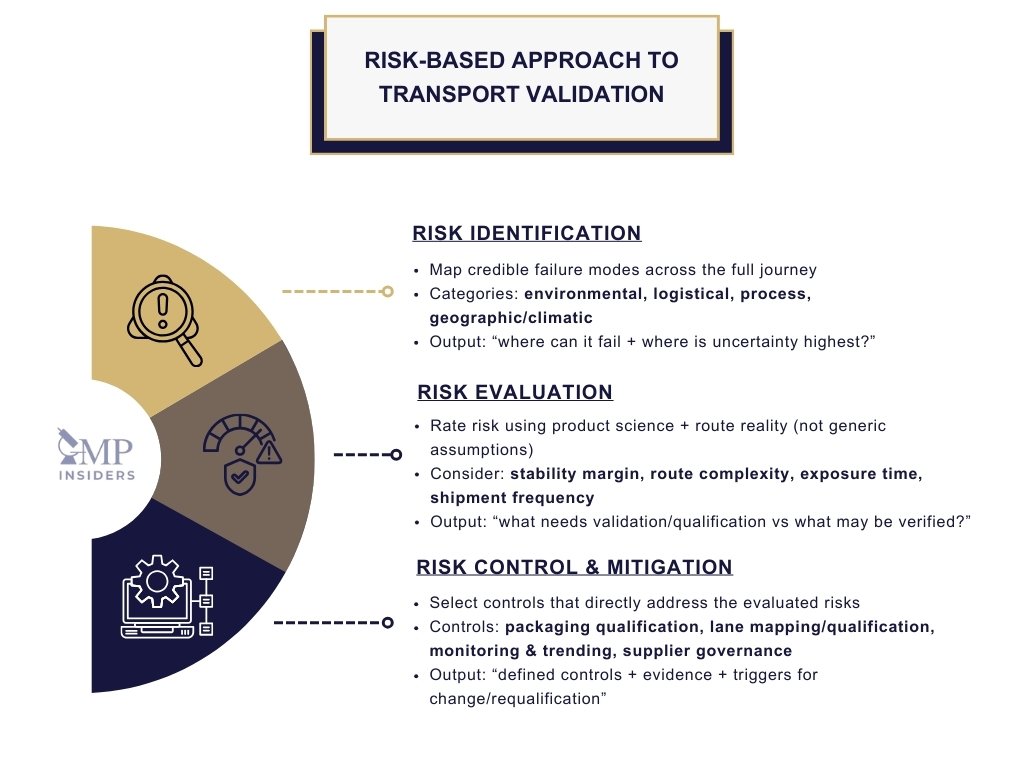
Core Principles of Risk-Based Transport Validation
A compliant risk-based approach to transport validation typically rests on three interconnected pillars. Together, they make the validation strategy defensible: it focuses effort where failure would matter, it scales evidence to risk, and it provides a clear scientific rationale for the chosen controls.
1) Risk Identification
This step is about mapping credible failure modes across the full distribution journey, not just “temperature control.” Typical risk categories include:
- Environmental risks: exposure to temperature extremes, uncontrolled humidity, light sensitivity (where relevant), and rapid transitions between environments (e.g., warm warehouse → cold truck).
- Logistical risks: delays at hubs or borders, missed connections, misrouting, prolonged tarmac time, or extended time in uncontrolled staging areas.
- Process risks: weak handovers between parties, unclear responsibilities, incomplete or inconsistent documentation, poor excursion handling, or gaps in communication when something deviates from plan.
- Geographical and climatic exposure: seasonal extremes, lane-specific climate patterns, altitude effects, and known hot/cold spots along the route (including last-mile handling).
The output of this step should be a structured risk picture: which hazards exist, where they can occur, and which parts of the lane drive the highest uncertainty.
2) Risk Evaluation
Once risks are identified, they must be evaluated using product science and route reality, not generic assumptions. This usually includes:
- Product vulnerability and stability margins: how close the product is to its limits (label storage conditions, excursion tolerance, stability data, sensitivity to short-term spikes).
- Route complexity and predictability: number of handovers, reliance on third parties, likelihood of unplanned storage, and the ability to intervene if conditions drift.
- Duration and exposure time: total transit time, expected dwell times, and the worst-case exposure scenarios (including weekends/holidays).
- Frequency of shipment: whether this is a one-off lane or a routine distribution that needs ongoing control, trending, and periodic review.
The goal is to determine where the true risk sits: a short route with high stability and strong controls may justify lighter evidence, while a long, variable lane with multiple handovers typically demands stronger qualification and ongoing oversight.
3) Risk Control and Mitigation
Risk-based validation is only credible if it results in concrete, effective controls. Controls should be selected to address the specific risks identified and evaluated, for example:
- Packaging selection and qualification: choosing packaging that can maintain required conditions under worst-case profiles, and qualifying it against realistic or conservative challenges.
- Lane qualification and mapping: understanding the lane’s thermal and logistical behavior, identifying weak points, and demonstrating that the lane is capable of maintaining the required conditions.
- Monitoring strategy: defining what is monitored, where, how often, with what alarm/alert limits, and how data is reviewed and trended (including excursion investigation requirements).
- Supplier qualification and governance: ensuring carriers, logistics providers, and warehouses are qualified, responsibilities are clear, quality agreements are in place, and performance is monitored through KPIs and periodic review.
When these three pillars are applied consistently, transport validation becomes intentional (based on known risks), proportional (evidence scaled to criticality), and scientifically justified (rooted in product stability and lane behavior), exactly what regulators expect to see.
What Needs to Be Considered in Transport Risk Assessment
A transport risk assessment is meaningful only when it moves beyond generic statements (“risk of temperature excursion”) and demonstrates that the company understands its actual distribution reality.
Key factors typically fall into four dimensions:
Product-Related Risks
These define how vulnerable the product is and how much “margin” exists before quality is impacted.
- Stability profile and sensitivity: labelled storage conditions, excursion tolerance (if defined), and known sensitivities (e.g., temperature, freeze/thaw, agitation, light).
- Degradation behavior: what changes when conditions drift (potency loss, impurity formation, physical instability) and whether degradation is gradual or can occur rapidly.
- Shelf life remaining during shipment: reduced remaining shelf life can reduce acceptable risk tolerance, especially for products close to expiry or with known stability constraints.
- Criticality of the medicine: life-saving or narrow-therapeutic-index products typically warrant stricter risk-control logic than lower-criticality products, even if storage conditions appear similar on paper.
Environmental Risks
These define the external stress profile to which the shipment is exposed, including seasonal and regional variability.
- Climatic zones: expected climatic exposure along the route, commonly aligned with ICH climatic zones (I–IV), where relevant for regional context and worst-case assumptions.
- Seasonal variation: summer vs winter stress, shoulder seasons, and short-term extreme events (e.g., heatwaves, cold snaps) that can meaningfully change lane behavior.
- Humidity exposure: relevant for moisture-sensitive products and for packaging integrity considerations (e.g., condensation, label damage, secondary packaging weakening).
- Potential for extreme temperatures: not only averages, consider peak exposure during staging, on-airport tarmac time, customs holds, and last-mile delivery.
Transport and Route Risks
These define how the shipment is moved and where the distribution chain is most likely to lose control.
- Mode of transport: road, air, or sea, and combinations, each with different risk profiles (e.g., air: tarmac + hub dwell risk; sea: prolonged duration and container variability).
- Duration and variability of transit time: nominal duration is less important than the credible range (best case vs worst case) and where time is spent (controlled vs uncontrolled).
- Number of handovers and control points: every transfer between parties increases the probability of mishandling, delays, and documentation gaps.
- Reliability of infrastructure: mature lanes may have predictable performance; developing markets may introduce higher variability in storage, power stability, and handling discipline.
- Known historical deviation patterns: previous temperature excursions, delays, repeated weak points, and seasonal trends should directly inform risk scoring and mitigation planning.
Operational and Process Risks
These define whether the quality system can actually execute and maintain control.
- Competence of logistics providers: training, qualification status, GDP maturity, deviation handling capability, and responsiveness.
- Availability and performance of packaging: whether qualified packaging is consistently available, correctly assembled, and fit for the lane and season.
- Monitoring capability: data logger strategy, calibration status, alarm logic, data retrieval, review timelines, and handling of missing data.
- Documentation discipline: completeness of shipping records, chain-of-custody evidence, excursion documentation, and traceability of decisions.
- Oversight responsibilities: clarity on who owns release decisions, who investigates excursions, who trends lane performance, and how governance is maintained across third parties.
A strong system can demonstrate through documented assessment outputs that each of these dimensions has been evaluated, that the risk conclusions are defensible, and that the resulting controls (qualification, monitoring, governance) are proportionate to the risk profile.
Transport Qualification Studies
Qualification studies are the evidence layer of transport validation. They convert the risk assessment and design assumptions into documented proof that the lane, packaging, monitoring, and operations can maintain required conditions under realistic (or justified worst-case) scenarios.

A strong program demonstrates three things: it was designed with intent, executed with discipline, and concluded with defensible criteria.
Study design: prove the right thing, on purpose
- Clear scope: what is being qualified (lane/route, packaging system, provider/transport process) and which product groups it applies to.
- Representative, risk-based selection: include relevant seasons, durations, and route complexity; use worst-case profiles where justified (e.g., hottest season, longest dwell time, most handovers).
- Justified number of runs: enough shipments to demonstrate repeatability; “single shipment” is rarely defensible unless risk is demonstrably low and documented.
- Defined expectation: what “success” looks like and why (linked to product stability data and risk assessment).
Monitoring: data quality determines study credibility
- Fit-for-purpose loggers: accuracy, range, battery, memory appropriate to the journey.
- Calibration controls: traceable calibration valid at time of use, documented intervals, certificates available.
- Placement strategy: deliberate locations based on risk (warm/cold points, thermal core vs edge, known vehicle/container hotspots), documented in the protocol.
- Data integrity: secure records, controlled access, audit trail where applicable, and retrievability for inspection.
Execution: reflect real life, under control
- Pre-approved protocol: no retrospective protocols; roles and responsibilities defined; QA oversight where appropriate.
- Operational realism: studies should mirror routine behavior, not “perfect-case” handling.
- Controlled documentation: time stamps, chain-of-custody, and transparent recording of deviations.
Acceptance criteria: defined upfront and scientifically justified
- Temperature/humidity performance: within specified limits, with excursion logic only where justified by stability data (not arbitrary).
- Repeatability: evidence that performance is consistent across shipments/seasons when needed.
- System integrity and practicality: packaging performance, process feasibility, and provider capability to execute consistently.
Reporting and deviation handling
A credible report summarizes what was done, presents clear data, evaluates results against acceptance criteria and stability rationale, and states a clear conclusion with limits of validity.
Deviations and outliers are recorded transparently, assessed for impact, investigated as needed, and trigger repeat studies or requalification only when scientifically justified (with a CAPA if systemic issues are found).
Regulatory Guidelines Relevant to Transport Validation
Transport validation requirements are not defined by a single regulation but are derived from a combination of GDP, GMP, WHO, pharmacopoeial, and risk-management frameworks. Collectively, these guidelines require companies to demonstrate that transportation conditions are controlled, scientifically justified, risk-based, and verifiable through documented evidence.
The most relevant frameworks include the following:
EU Guidelines on Good Distribution Practice (GDP)
The EU GDP guideline is the primary regulatory driver for transport validation in Europe. It establishes that medicinal products must be stored and transported under conditions that do not compromise quality.
Key reference:
Core expectations:
- Transport conditions must comply with labeled storage requirements
- Transportation systems, routes, and packaging should be qualified or validated
- Temperature-sensitive products require appropriate monitoring and evidence of control
- Responsibilities between manufacturers, wholesalers, and logistics providers must be clearly defined
- Records must demonstrate compliance and traceability
EU GMP – Annex 15 (Qualification and Validation)
Although Annex 15 focuses on manufacturing systems, its validation philosophy applies directly to transport.
Key reference:
Relevant expectations:
- Systems must be proven fit for their intended use.
- Validation and qualification must follow structured lifecycle thinking
- Risk-based approaches must define scope and intensity of validation
- Activities must be scientifically justified, documented, and periodically reviewed
WHO Guidance
WHO places strong emphasis on protecting temperature-sensitive pharmaceuticals during global distribution. Its guidance is widely referenced internationally, especially beyond the EU.
Key references:
- WHO Technical Report Series 961, Annex 9 – Model Guidance for Storage & Transport of Time- and Temperature-Sensitive Pharmaceutical Products
- WHO Good Storage and Distribution Practices – Technical Report Series 1025, Annex 7
Key expectations:
- Transport environments must be understood, qualified, and controlled
- Packaging/insulated shipping systems require performance qualification
- Monitoring devices must be calibrated and reliable
- Risk assessments must consider climatic zones, duration, and route complexity
- Documentation must clearly demonstrate control throughout transport
Pharmacopoeial and US Expectations
While not always prescriptive on methodology, pharmacopoeial standards reinforce that product quality must be protected during distribution.
Key references:
- USP <1079> Good Storage and Distribution Practices
- USP <1079.2> Mean Kinetic Temperature in the Evaluation of Temperature Excursions During Storage and Transportation of Drug ProductsPackaging and Transport for Temperature-Sensitive Products
They emphasize:
- Alignment of transport controls with stability data
- Maintaining integrity of time- and temperature-sensitive products (TTSPPs)
- Proper packaging qualification and environmental monitoring strategies
FAQ
Do Regulators Expect Transport Validation for Every Product and Every Lane?
No, regulators do not expect a blanket approach where every transport scenario is fully validated. What they expect is a risk-based decision-making process supported by documented rationale.
If validation is not performed, the organisation must demonstrate why it was not necessary. The decision must reflect product stability, route complexity, and historical performance. What matters most to inspectors is logic, transparency, and defensibility.
Is It Acceptable to Categorize Transport Lanes Instead of Validating Each Individually?
Yes, lane categorization is widely accepted when it is scientifically justified. Organisations may group lanes based on climate similarity, duration, infrastructure maturity, and transport modality. Once grouped, representative or worst-case lanes can be qualified.
However, the justification for grouping must be explicit, documented, and periodically reassessed. Inspectors expect evidence that similarities are real, not assumed.
What Is the Role of the Responsible Person (RP) in Transport Validation?
The RP is expected to have oversight of transport validation as part of the GDP responsibility. They must understand how validation was designed, what conclusions it supports, and how ongoing assurance is maintained.
The RP typically approves key validation documentation and excursion-related decisions. They must be able to confidently explain the rationale to inspectors. Ultimately, they help ensure transport validation is not a technical exercise disconnected from governance.
How Should Companies Handle Situations Where Validation Results Are Borderline?
Borderline results require scientific discussion rather than automatic acceptance or rejection. Organisations should analyze whether risks are justified, controlled, and explainable. Stability impact assessment is essential.
If residual uncertainty remains, additional mitigation such as packaging reinforcement or enhanced monitoring may be required. Documented justification and transparency are key.
Does Transport Validation Apply to Investigational Medicinal Products (IMPs)?
Yes, it applies, though expectations are applied proportionally. Clinical trial supplies must be protected with the same scientific integrity as commercial products. Risk-based justification remains essential. The decision must consider product sensitivity and patient impact. Authorities reviewing clinical trial distribution often scrutinize transport practices closely.
How Does Transport Validation Integrate With Product Release?
Transport validation provides supporting assurance for release decision-making. It demonstrates that distribution conditions are controlled as part of the overall quality system.
When deviations occur, historical validation evidence strengthens scientific assessment. RP and QA rely on validation records to determine acceptability. Without validation, release justification becomes significantly weaker.
Final Thoughts
Transport validation is not a side topic in GDP; it is a direct extension of GMP thinking into the real world where products actually move. Once a batch leaves a controlled warehouse, its quality remains the responsibility of the manufacturer or marketing authorization holder, and regulators increasingly expect clear, defensible evidence that this responsibility is taken seriously throughout the distribution chain.
What differentiates a strong program from a weak one is not the number of documents, but the coherence of the story they tell: from risk assessment through DQ/OQ/PQ to routine monitoring and CAPA-driven improvement. When that story is logical, science-based, and reflected in day-to-day practice, transport becomes a controlled quality process rather than a variable logistics risk.
Ultimately, transport validation is about confidence: confidence that products arrive in the same state in which they were released, confidence that inspection questions can be answered with data and rationale, and confidence that the organisation understands its supply chain well enough to defend it.















