Cleanrooms are a critical component of pharmaceutical manufacturing, mainly when producing sterile or high-risk medicinal products. To maintain product safety and comply with global regulatory expectations, manufacturers must control airborne particulate contamination by designing and operating facilities within defined cleanliness standards.
One of the most widely accepted frameworks for cleanroom classification is the ISO 14644-1 standard, which outlines specific limits for airborne particles based on size and concentration.
Related Article: Pharmaceutical Cleanrooms in GMP
Regulatory systems like the EU GMP Annex 1 and FDA aseptic processing guidance incorporate ISO-based classifications but add further microbiological and procedural requirements.
This article breaks down the ISO cleanroom classes, particle count requirements, and how they connect to EU GMP Annex 1 Grades A through D, and the key considerations companies must address to ensure compliance throughout facility design, operation, and monitoring.
ISO 14644-1 is the foundational international standard that defines the classification of air cleanliness in cleanrooms and controlled environments, based on the concentration of airborne particles.
Originally developed to replace the outdated FED-STD-209E (Airborne Particulate Cleanliness Classes in Cleanrooms and Clean Zones), it is now globally recognized and applied across industries, with particular relevance in pharmaceuticals, biotechnology, and medical device manufacturing.
ISO 14644-1 establishes nine cleanroom classes, from ISO Class 1 (the cleanest) to ISO Class 9 (the least clean), which is determined by the maximum allowable particle concentration per cubic meter of air for specific particle sizes (≥0.1 μm, ≥0.3 μm, ≥0.5 μm, etc.).
Pharmaceutical manufacturers typically operate in ISO Classes 5, 7, and 8, as these correspond to the cleanroom grades (A–D) defined in EU GMP Annex 1. Each classification is determined by:
The classification must be validated at rest and, where required, in operation, depending on the criticality of the area. ISO 14644-1 forms the basis for system design and environmental control strategies, but compliance with it alone does not guarantee GMP conformance.
Instead, it provides the baseline that must be integrated with operational procedures, cleaning validation, and microbial monitoring.
ISO 5 is among the strictest cleanroom classifications commonly used in pharmaceuticals.
Related Article: ISO Class 5 Cleanroom Requirements
ISO 6 is less common in pharmaceutical facilities but can serve as a transition or background environment.
ISO 7 is one of the most widely used classifications in sterile manufacturing.
ISO 8 is the most common classification for support and preparation areas in pharma.
ISO 9 is the least strict classification under ISO 14644-1.
It’s essential to differentiate between:
ISO 14644-2 governs ongoing monitoring, while ISO 14644-1 governs initial classification.
Cleanroom classification under ISO 14644-1 is based on quantitative measurement of airborne particulate matter. These classifications define the maximum allowable concentration of particles per cubic meter for specified size ranges and serve as a baseline for cleanroom design, operation, and qualification.
The primary determinant of classification is the concentration of airborne particles equal to or greater than specific sizes, typically:
Pharmaceutical applications most commonly monitor particles ≥0.5 μm and ≥5.0 μm, as these sizes are relevant to product contamination and serve as reference points in EU GMP Annex 1.
Each ISO Class specifies the maximum allowable particle concentration (in particles per cubic meter).
(Particles per cubic meter of air)
| ISO Class | ≥0.1 µm | ≥0.2 µm | ≥0.3 µm | ≥0.5 µm | ≥1.0 µm | ≥5.0 µm |
|---|---|---|---|---|---|---|
| ISO 1 | 10 | — | — | — | — | — |
| ISO 2 | 100 | 24 | 10 | — | — | — |
| ISO 3 | 1,000 | 237 | 102 | 35 | 8 | — |
| ISO 4 | 10,000 | 2,370 | 1,020 | 352 | 83 | — |
| ISO 5 | 100,000 | 23,700 | 10,200 | 3,520 | 832 | 29 |
| ISO 6 | — | — | — | 35,200 | 8,320 | 293 |
| ISO 7 | — | — | — | 352,000 | 83,200 | 2,930 |
| ISO 8 | — | — | — | 3,520,000 | 832,000 | 29,300 |
| ISO 9 | — | — | — | 35,200,000 | 8,320,000 | 293,000 |
This data forms the benchmark during cleanroom qualification and is verified through particle counting during operational states (at rest/in operation).
To classify a cleanroom or clean zone, the following steps are required:
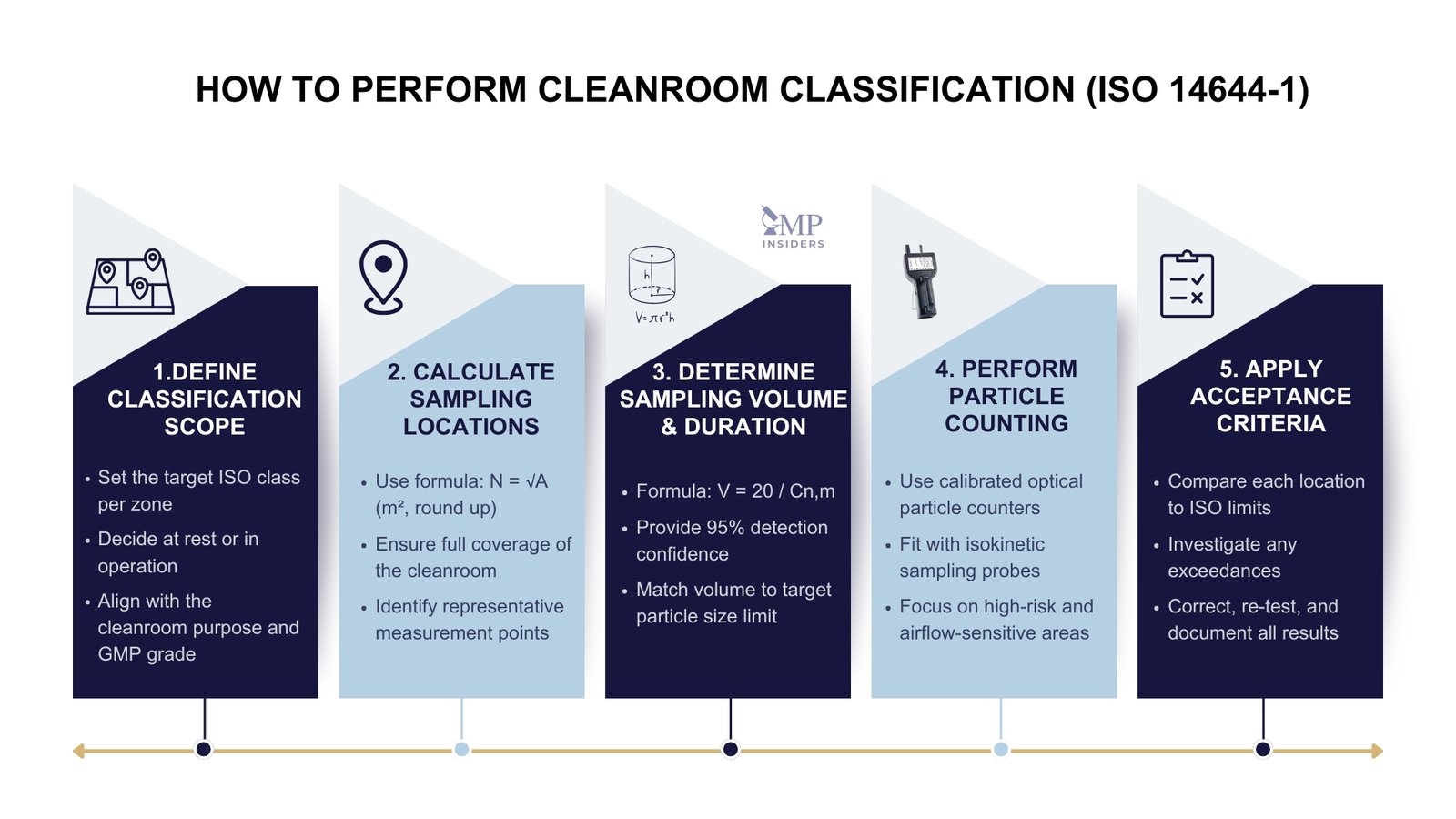
Sampling must be carried out using calibrated optical particle counters fitted with isokinetic sampling probes to ensure representative collection of airborne particles. Sampling locations should be those identified during the planning phase, focusing on areas at highest risk of contamination. These typically include:
Measurements should be taken without disturbing normal airflow conditions. For in operation classifications, sampling must reflect typical operational activities to provide an accurate assessment of cleanroom performance under real working conditions.
Results from each sampling location are compared against the ISO 14644-1 particle concentration limits for the target class. All measured values must be within the specified limits for the cleanroom to achieve classification. If one or more locations exceed the allowable concentration:
All results, calculations, and justifications must be documented as part of the cleanroom qualification records, following GMP documentation requirements.
Pharmaceutical manufacturers must comply with both technical standards (ISO 14644-1) and regulatory guidelines (EU GMP Annex 1).
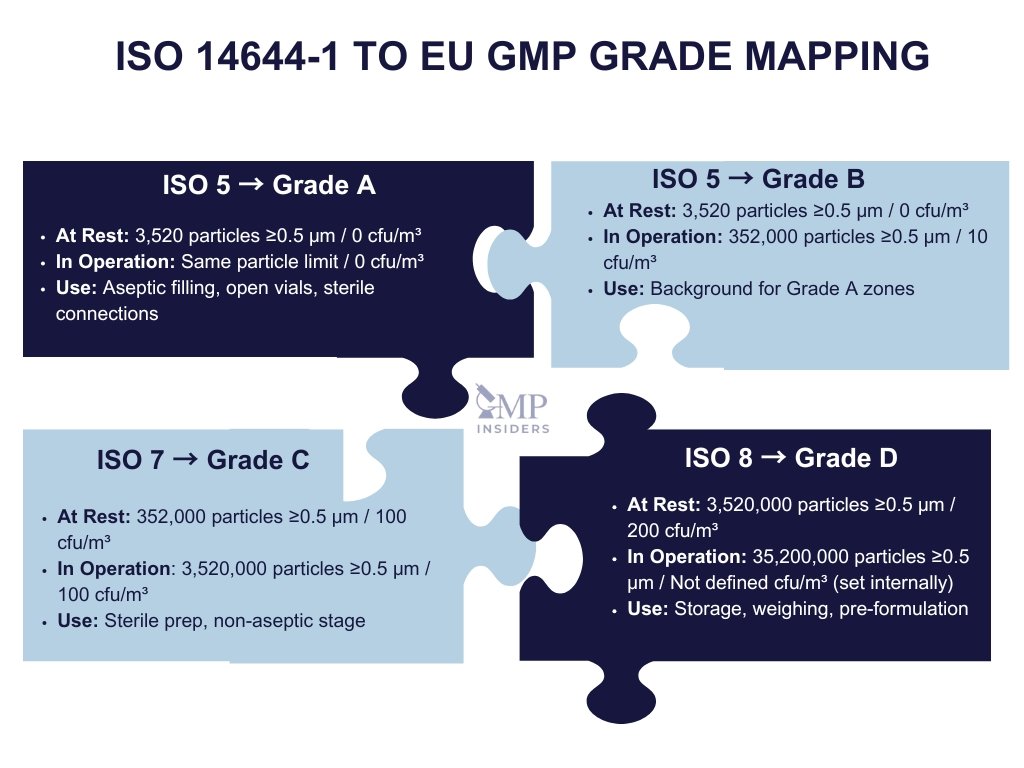
While ISO 14644-1 defines air cleanliness in terms of airborne particles, Annex 1 introduces GMP Grades that incorporate not only particle limits but also microbiological requirements and operational risk levels.
This mapping is critical for:
SEE ALSO: GMP Cleanroom Classifications: Grade A, B, C, D
| GMP Grade | ISO Class (at rest) | ISO Class (in operation) | Common Use Areas | Microbial Limits (cfu/m³) |
|---|---|---|---|---|
| Grade A | ISO 5 | ISO 5 | Aseptic filling, open vials, sterile connections | 0 |
| Grade B | ISO 5 | ISO 7 | Background to Grade A zone (aseptic core support) | 10 |
| Grade C | ISO 7 | ISO 8 | Preparation of sterile products (non-aseptic stage) | 100 |
| Grade D | ISO 8 | Not defined* | Storage, component weighing, pre-formulation | 200 |
*Note: Annex 1 defines Grade D only “at rest” — operational limits may be set internally based on risk assessment.
While ISO 14644-1 provides limits for non-viable particles, EU GMP Annex 1 supplements this with viable microbiological limits, which are assessed using:
Aseptic Filling Line (Open Vial Fill)
SEE ALSO: Media Fill Tests – Aseptic Process Simulation (APS)
Cleanroom for Sterile API Packaging
Terminally Sterilized Product Manufacturing
Before a cleanroom can be used for pharmaceutical manufacturing, it must undergo a formal qualification process to confirm that its design, installation, and operation meet the required ISO class and GMP grade.
Cleanroom qualification is typically performed in three phases:
The qualification must be documented in detailed protocols and executed using calibrated instruments and validated test methods.
SEE MORE: Cleanroom Qualification and Validation
Per ISO 14644-3, the following performance tests are typically performed:
| Test Type | Purpose | Frequency |
|---|---|---|
| Airborne particle count | To confirm ISO classification | Required for initial and periodic qualification |
| HEPA filter integrity | To ensure filters are leak-free (e.g., PAO/DOP test) | At installation and periodically |
| Airflow velocity and volume | To verify laminarity and room pressurization | Initially and as needed |
| Airflow visualization (smoke study) | To confirm unidirectional flow and absence of turbulence | At installation and for media fills |
| Pressure differentials | To confirm pressure cascades between grades | Continuous or periodic monitoring |
| Temperature and RH | To ensure comfort and compliance | Continuous or routine |
All qualification data must be reviewed and approved by QA before release for GMP operations.
Once qualified, cleanrooms require a routine monitoring program to demonstrate continued compliance and to detect any environmental deterioration that may affect product quality.

EMP includes both non-viable particulate and viable microbiological monitoring, based on:
Pharmaceutical companies must define:
Deviations beyond these levels must be investigated via deviation management and may lead to hold or rejection of associated batches.
SEE ALSO: Environmental Monitoring In the Pharmaceutical Industry
Environmental data must not only be collected but also analyzed over time to identify:
Tools such as statistical trend charts, heat maps, and Pareto analyses support a risk-based contamination control strategy.
ISO 14644-2 requires periodic requalification:
Requalification intervals must be justified and documented as part of the facility’s contamination control strategy and quality system.
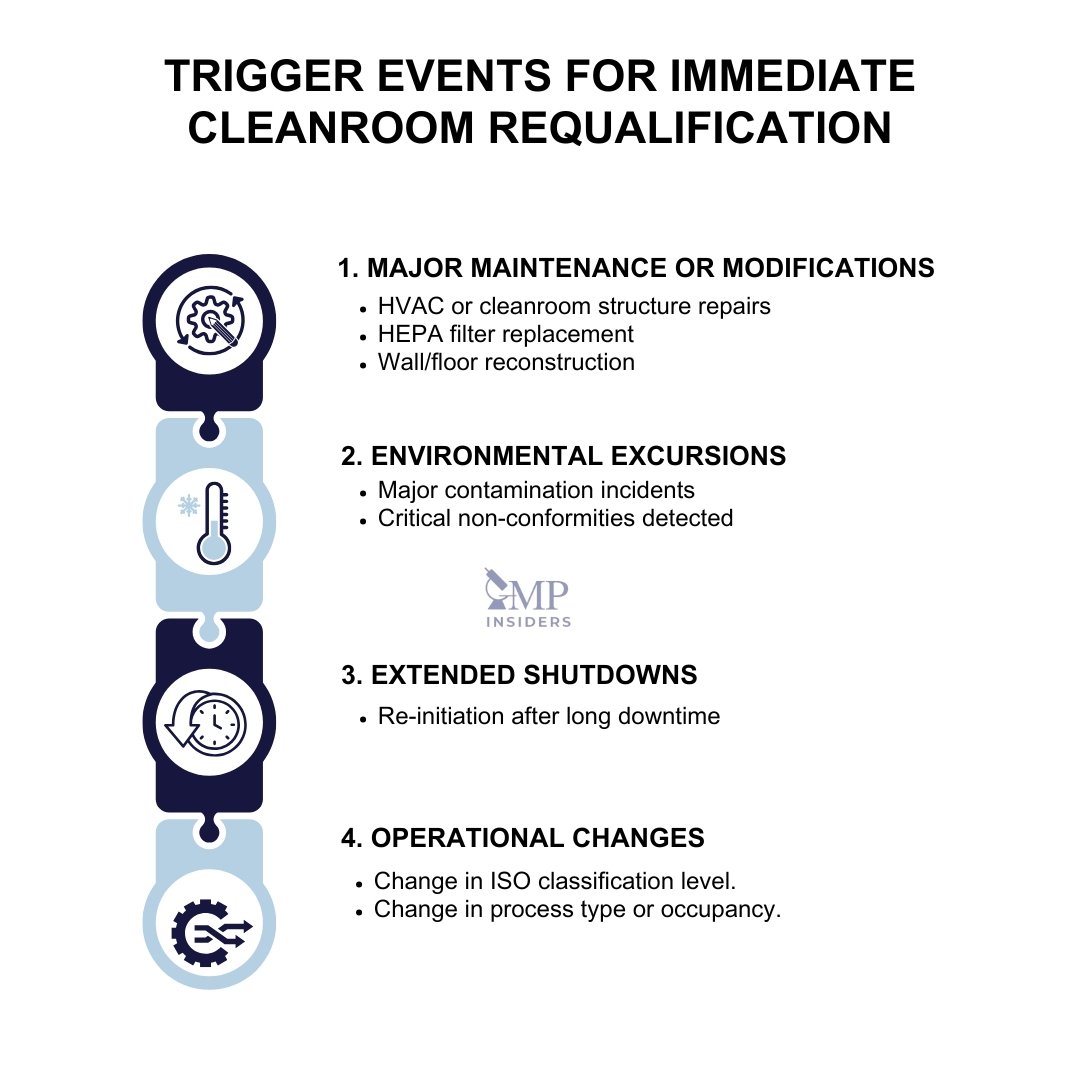
Requalification should be performed outside of the routine schedule if any of the following occur:
All requalification activities must include:
Below are some of the ISO 14644 series and related standards that support the full lifecycle management of cleanroom performance in GMP pharmaceutical settings.
ISO 14644-2 complements ISO 14644-1 by providing guidance on the ongoing monitoring of cleanrooms to ensure that they continue to meet their classification requirements over time.
Key Concepts:
Pharmaceutical manufacturers typically integrate these recommendations into their Environmental Monitoring Programs (EMP) and requalification protocols.
ISO 14644-3 specifies test methods and procedures used during cleanroom qualification and requalification.
Common Tests Include:
This standard is essential for executing IQ/OQ/PQ in GMP cleanroom projects and is often cross-referenced in validation protocols and engineering acceptance testing (FAT/SAT).
The ISO 14644-4 specifies requirements for designing, constructing, and commissioning cleanrooms and clean air devices. It covers facility layout, HVAC specification, materials, zoning, and operational commissioning protocols.
Widely applied in pharmaceutical facility design to ensure the operational environment supports ISO cleanliness targets from the start.
A major revision of the ISO 14644-5 was released in mid-2025 redefining cleanroom operations to include a formal Operations Control Programme (OCP). The OCP requires documented procedures for:
The updated standard integrates various industry Recommended Practices (e.g. IEST-RP‑CC003–5) and aligns more closely with modern pharmaceutical regulatory expectations and cleanliness frameworks.
Published in 2019, ISO 14644-16 addresses the design, operation, and maintenance of cleanrooms to energy efficiency, a growing concern for sustainability in pharmaceutical manufacturing.
Why it Matters:
Although not part of the 14644 series, ISO 14698 is frequently referenced in pharmaceutical contexts, particularly where aseptic processing or biologics are involved.
Key Elements:
While EU GMP Annex 1 is now the primary source for microbiological expectations, ISO 14698 remains useful for building robust contamination control frameworks.
SEE ALSO: Risk-based Contamination Control Strategy
Yes, it’s common for cleanrooms to have areas or zones classified to different ISO levels depending on the level of contamination risk. For example, an ISO 5 LAF zone used for aseptic filling may be located within an ISO 7 background room. Zonal classification supports effective contamination control while allowing operational flexibility and energy efficiency.
ISO Classification is performed using calibrated optical particle counters (OPCs) capable of detecting particles down to 0.5 µm or smaller. These instruments must be used with isokinetic probes and placed at pre-determined sampling locations. Some facilities also use automated monitoring systems with data logging for ongoing performance validation.
Yes. Localized clean air devices like isolators, LAF hoods, and RABS must be classified under ISO 14644-1 using the same airborne particle criteria. Because these devices are often used in aseptic operations, they must achieve ISO 5 under dynamic conditions, with strict control over airflow, personnel interaction, and cleaning procedures.
No, ISO 14644 does not define fixed air change rates. Instead, it states that the room must maintain particle concentrations within the required limits. However, typical ranges are widely accepted (e.g., 240 ACH for ISO 5, 30–60 ACH for ISO 7/8), and HVAC systems must be validated to support the required cleanliness level.
Cleanroom classification according to ISO 14644-1 forms the technical foundation for maintaining controlled environments in pharmaceutical manufacturing. However, compliance with ISO particle limits alone is not sufficient.
Proper control requires the integration of classification, environmental monitoring, personnel behavior, facility design, and microbiological safeguards, all aligned with regulatory frameworks such as EU GMP Annex 1.
By understanding how ISO classes map to GMP grades and implementing a risk-based environmental monitoring strategy, manufacturers can ensure their cleanrooms remain in a state of control throughout their lifecycle.