ISO 7 cleanrooms are a key component of pharmaceutical facilities designed for the manufacture of sterile medicinal products. These areas provide the controlled background environment necessary to support higher-classified critical zones and to reduce the risk of contamination during operations where product protection is required.
Within sterile manufacturing facilities, ISO 7 areas are commonly used alongside operations performed under ISO 5 conditions and, depending on the facility design and operational state, may correspond to the environmental expectations for Grade B background areas at rest. Their function is not limited to spatial separation. They contribute directly to maintaining appropriate airborne cleanliness, pressure differentials, and environmental conditions required to support aseptic processing.
In contrast to ISO 5 zones, which are characterized by unidirectional airflow and highly localized protection of critical points, ISO 7 cleanrooms generally operate with non-unidirectional airflow and are subject to a broader range of operational influences, including personnel presence, material transfer, equipment configuration, and HVAC performance.
For this reason, the design, qualification, monitoring, and routine operation of ISO 7 cleanrooms are critical to the overall contamination control strategy. Inadequate control of these areas may compromise the performance of adjacent critical zones and weaken the effectiveness of the measures implemented to assure sterility.
What Is an ISO 7 Cleanroom?
An ISO 7 cleanroom is a classified environment defined in ISO 14644-1 within the broader framework for airborne particulate cleanliness. It represents an intermediate level of environmental control within the ISO classification system and is commonly applied in areas where contamination must be controlled but where the highest level of localized protection is not required throughout the entire room.
In pharmaceutical and biotechnology facilities, ISO 7 cleanrooms are frequently used as the background environment for operations performed under more critical conditions. Their airborne particle limits are more stringent than those applicable to ISO 8 areas, while remaining less restrictive than those assigned to ISO 5 zones used for critical aseptic exposure. As a result, ISO 7 occupies an important position in the overall contamination control strategy, particularly where adjacent higher-classified areas depend on a stable and well-controlled surrounding environment.
In sterile manufacturing applications, ISO 7 conditions are often associated with areas supporting Grade A operations and, depending on facility design and operational state, may correspond to the expectations for Grade B conditions at rest.
In this function, the cleanroom must provide a level of control sufficient to limit the transfer of contamination toward critical zones while still permitting routine operational activities, such as personnel movement, equipment placement, and material handling, under defined conditions.
See Also: Pharmaceutical Cleanrooms in GMP: Definition, Design and More…
Because ISO 7 areas are often located immediately adjacent to critical aseptic processing zones, their performance directly influences those zones’ ability to remain in a state of control. For this reason, ISO 7 cleanrooms should be regarded as an integral part of sterile manufacturing control rather than as a secondary background space.
Position of ISO 7 in the ISO 1–9 Classification Scheme

Within the ISO 14644-1 classification system, ISO 7 occupies an intermediate position between the highest levels of particulate control and environments with lower cleanliness requirements. The sequence of classification extends from ISO 1, representing the most stringent airborne particulate control, to ISO 9, which approximates the particulate conditions of ambient air.
ISO 7, therefore, provides a level of control that is lower than ISO 5 and ISO 6, but significantly higher than ISO 8 and unclassified areas. This distinction is important in facility design because each classification supports a different type of manufacturing activity and level of product protection.
In practice:
- ISO 5 = aseptic exposure
- ISO 7 = aseptic background
- ISO 8 = preparation/wash/staging
| Feature | ISO 5 (Grade A) | ISO 7 (Grade B/Background) | ISO 8 (Grade C/D) |
|---|---|---|---|
| Primary Role | Critical Aseptic Exposure | Aseptic Background/Support | Prep, Wash, & Staging |
| Particle Limits | Most Stringent | Intermediate | Least Restrictive |
| Common Activities | Filling, Sealing, Open Product | Gowning, Buffer Prep, Corridors | Component Washing, Staging |
| Typical Airflow | Unidirectional (Laminar) | Non-unidirectional (Turbulent) | Non-unidirectional |
| Proximity to Product | Direct Contact | Adjacent/Support | Indirect/Upstream |
Where ISO 7 Cleanrooms Are Commonly Used
ISO 7 cleanrooms are used in sectors where a controlled environment is required to reduce the risk of particulate contamination and to support operations performed under more critical conditions. Their application is particularly common in facilities where the surrounding environment must remain under defined control, even when full ISO 5 conditions are not required throughout the room.
Typical uses include the following:
Pharmaceutical and biotechnology facilities
- Background areas for aseptic filling lines
- Preparation and staging areas for sterile components
- Aseptic gowning rooms
- Buffer and solution preparation rooms
- Clean transfer corridors supporting higher-classified areas
Medical device manufacturing
- Packaging of clean or contamination-sensitive components
- Assembly areas for devices requiring controlled environmental conditions
Microelectronics and optics
- Assembly of sensors, imaging components, and other particulate-sensitive equipment
ATMP, cell therapy, and gene therapy facilities
- Pre-processing and support spaces positioned upstream of Grade A or Grade B critical operations
ISO 7 serves as a controlled environment that supports process protection and reduces the risk of contamination transfer to more critical steps. Its importance becomes particularly pronounced when the next stage of the operation involves sterile exposure, critical product contact, or another activity requiring heightened environmental control.
Class ISO 7 Cleanrooms Particle Count Limits
ISO 7 classification is defined by the maximum allowable concentrations of airborne non-viable particles under ISO 14644-1. These limits determine whether a room has sufficient filtration performance, airflow stability, and environmental control to support high-risk operations such as aseptic filling or sterile staging.
Although ISO 7 is less stringent than ISO 5, it still represents a controlled environment with substantially tighter particulate limits than ISO 8 or unclassified areas. Its performance is therefore significant not only in relation to its own classification status, but also in terms of its effect on adjacent critical zones.
When ISO 7 areas are used as the background for aseptic processing, loss of control may increase the risk of cross-contamination during routine operations, interventions, or material movement.
ISO 7 Non-Viable Particle Limits (ISO 14644 Definition)
ISO 7 allows higher particle levels than ISO 5 or ISO 6, but still requires strong airflow control.
Maximum permitted particles per m³:
| Particle size threshold | Maximum permitted particles per m³ | Practical meaning |
|---|---|---|
| ≥ 0.5 µm | 352,000 | Main reference point for confirming ISO 7 airborne particulate cleanliness |
| ≥ 1.0 µm | 83,200 | Supports evaluation of overall cleanliness and filtration performance |
| ≥ 5.0 µm | 2,930 | Indicates control of larger airborne particles that may reflect airflow disturbance or contamination events |
These limits apply to the classification of the cleanroom and provide the basis for confirming that the room meets the required level of airborne particulate cleanliness.
From an operational perspective, these limits indicate that:
- ISO 7 permits a substantially higher particle concentration than ISO 5
- HEPA-filtered supply air remains necessary to achieve and maintain classification
- Non-unidirectional airflow may be acceptable, provided that its performance is appropriately demonstrated
- Air change rates and airflow patterns must be sufficient to support effective particle removal
- The room design and HVAC system must consistently maintain the required state of control
At-Rest vs Operational Classification in ISO 7

ISO 14644 defines two testing conditions:
At-rest Condition
- Equipment is installed and operating in the agreed manner
- No personnel are present in the room
- This condition is typically used to assess the effectiveness of the HVAC system, airflow pattern, and basic particulate control capability
Operational Condition
- Personnel are present, and routine activities are being performed
- Particle generation increases due to operator movement, material handling, and equipment interaction
- This condition provides a more representative assessment of cleanroom performance during actual use
In practice, maintenance of ISO 7 conditions during operation depends on a combination of facility design, personnel discipline, material flow control, and environmental management. For this reason, cleanroom classification alone is not sufficient; ongoing performance verification remains necessary.
Monitoring Frequency (Non-Viable Particles)
In contrast to ISO 5 critical zones, ISO 7 cleanrooms do not generally rely on continuous monitoring of non-viable particles. The monitoring approach should instead be defined based on risk, room function, and the nature of the operation being supported.
Typical monitoring practices may include:
- Periodic non-viable particle sampling during routine operations
- Additional monitoring during activities associated with elevated contamination risk
- Monitoring following cleaning, maintenance, or other events that may affect the room status
- Sampling during periods of increased personnel activity or operational change
- Trend evaluation to identify deterioration in performance over time
| Monitoring situation | Typical purpose |
|---|---|
| Routine periodic sampling during operation | Confirms ongoing particulate control during normal use |
| Additional sampling during higher-risk activities | Detects increased contamination risk during interventions or unusual activity |
| Monitoring after cleaning or maintenance | Verifies that the room has returned to an acceptable controlled state |
| Sampling during periods of increased personnel activity | Assesses the effect of occupancy and movement on airborne particle levels |
| Trend evaluation over time | Identifies gradual deterioration, instability, or recurring loss of control |
The purpose of this approach is to confirm that the cleanroom remains in a controlled state on a sustained basis. In this context, monitoring is not limited to demonstrating compliance at a single point in time but should also support the detection of adverse trends, loss of stability, or conditions that may affect the surrounding aseptic environment.
Environmental Monitoring in ISO 7 Cleanrooms
Environmental monitoring in ISO 7 cleanrooms is an essential element of contamination control where these areas support sterile manufacturing operations. When an ISO 7 room serves as the surrounding environment for higher-classified zones, its state of control directly affects the protection of adjacent critical processing areas.

For this reason, environmental monitoring should be established as part of the overall contamination control strategy and used to confirm that the cleanroom remains in a qualified and operationally controlled state.
Two regulatory frameworks are relevant in this context:
- ISO 14644-1, which defines cleanroom classification on the basis of airborne particle concentration.
- ISO 14644-2, which specifies minimum requirements for a monitoring plan to demonstrate continued cleanroom performance in relation to particulate cleanliness.
- EU GMP Annex 1, which defines routine expectations for total particle monitoring, microbial monitoring, alert and action levels, and trend evaluation in sterile manufacturing environments.
| Framework | Main focus | Relevance to ISO 7 environmental monitoring |
|---|---|---|
| ISO 14644-1 | Cleanroom classification based on airborne particle concentration | Defines the particulate cleanliness class of the room |
| ISO 14644-2 | Ongoing monitoring plan for continued particulate performance | Requires a monitoring approach that demonstrates continued compliance over time |
| EU GMP Annex 1 | Routine total particle monitoring, microbial monitoring, alert/action levels, and trending in sterile manufacturing | Defines GMP expectations for environmental monitoring where the room supports sterile operations |
A sound understanding of how these requirements interact is important, particularly because ISO classification and GMP grading are related but not interchangeable concepts. ISO 14644 addresses airborne particulate cleanliness; it does not establish viable monitoring limits or characterize the microbiological state of the cleanroom. Those expectations arise from GMP requirements and the specific function assigned to the room within the manufacturing process
Non-Viable Particle Monitoring in ISO 7 Cleanrooms
The non-viable particle monitoring strategy for an ISO 7 cleanroom should be determined according to its operational use, the activities performed, and its relationship to critical zones. Where the room directly supports aseptic processing, the monitoring program should be capable of detecting increases in contamination levels and evidence of system deterioration in sufficient time for appropriate action.
Annex 1 requires continuous total particle monitoring for Grade A areas and recommends a similar approach for Grade B, although at a reduced sample frequency where justified. For Grade C and D areas, the monitoring strategy should be based on qualification data, routine results, and quality risk management.
See Also: GMP Cleanroom Classification According to Annex 1
Accordingly, a non-viable monitoring program for ISO 7 areas may include:
- routine sampling during operation at locations justified by risk assessment,
- additional monitoring in areas with high personnel or material activity,
- focused sampling near transfer interfaces and pathways to critical zones,
- monitoring during simulated operations where relevant,
- increased sampling following interventions, shutdowns, maintenance, or adverse events.
The purpose of this monitoring is not merely to confirm that individual results remain below a numerical limit. It is to provide meaningful information on the stability of the cleanroom environment over time and to support timely detection of deterioration in airflow control, filtration performance, or operational discipline.
Viable Environmental Monitoring in ISO 7
Viable monitoring in ISO 7 cleanrooms should be defined based on the GMP grade assigned to the room and the risk posed by the operations performed within it. Where aseptic operations are carried out, Annex 1 expects frequent microbial monitoring using a combination of methods, with the selection of methods justified within the contamination control strategy and demonstrated not to compromise the process or airflow pattern.
Common viable monitoring methods:
- Active air sampling (typical volumes: 0.5 m³ or 1 m³)
- Settle plates placed at strategic high-risk locations
- Contact plates on benches, equipment surfaces, and walls
- Personnel monitoring (gloves, gown surfaces), especially in ISO 7 gowning areas
Typical monitoring frequency:
- Daily, if ISO 7 cleanrooms directly supports aseptic filling
- Shift-based, in high-risk operations
- Weekly, if used as a lower-risk staging or transfer area
- Before and after ISO 5 operations, ensuring environmental reset
Annex 1 encourages a frequency that reflects occupancy and process criticality.
Alert and Action Levels for ISO 7 (Viable Monitoring)
Unlike ISO 5/Grade A, which sets action limits at 0 CFU, ISO 7 allows higher microbiological levels and is defined by risk and historical data.
Typical facility-defined limits (example ranges):
- Air samples: 5–10 CFU/m³
- Settle plates (4 hours): 5–15 CFU
- Contact plates: 3–10 CFU
- Glove prints: 1–3 CFU
These are example ranges: each facility must define its own limits based on trend data, process criticality, and Annex 1 recommendations for Grade B/C environments.
Routine environmental control should not rely solely on action limits. Annex 1 requires both alert levels and action limits to be established and periodically reviewed in light of qualification data and ongoing trend results.
Exceedance of an action limit should trigger a documented root-cause investigation, a product-impact assessment, and appropriate corrective and preventive action. Trend analysis remains particularly important in background cleanrooms because deterioration is often first detected through repeated minor shifts rather than a single isolated result.
Role of ISO 7 EM in Supporting ISO 5 Stability
Where an ISO 7 cleanroom forms part of the background environment for higher-classified operations, its environmental monitoring program should be designed to demonstrate continued control of the surrounding area and to provide early warning of conditions that could affect aseptic processing.
In this context, monitoring data serves a broader purpose than routine compliance. They provide evidence that the cleanroom continues to support the intended state of control for the process, the facility, and the wider contamination control strategy.
Stable ISO 7 EM performance directly supports:
- Reduced non-viable spikes in ISO 5
- Lower microbial load entering Grade A areas
- Faster recovery times after ISO 5 interventions
- Fewer EM investigations during aseptic runs
- Stronger contamination control strategy overall
Related Article: ISO 5 Cleanroom Requirements
Airflow and HVAC Requirements in ISO 7 Cleanrooms
Airflow control and HVAC performance are fundamental to an ISO 7 cleanroom’s ability to maintain its classified state during operation. In contrast to ISO 5 critical zones, which rely on unidirectional airflow to protect exposed product and critical surfaces at the working position, ISO 7 rooms are generally designed to operate with non-unidirectional airflow.
In these areas, the primary control mechanism is dilution and removal of contamination through an appropriately filtered air supply, suitable air distribution, and effective extraction. Where ISO 7 areas support Grade A or Grade B operations, the ventilation system should be capable of maintaining environmental stability under all operational conditions, including normal occupancy, material transfer, and routine equipment use.
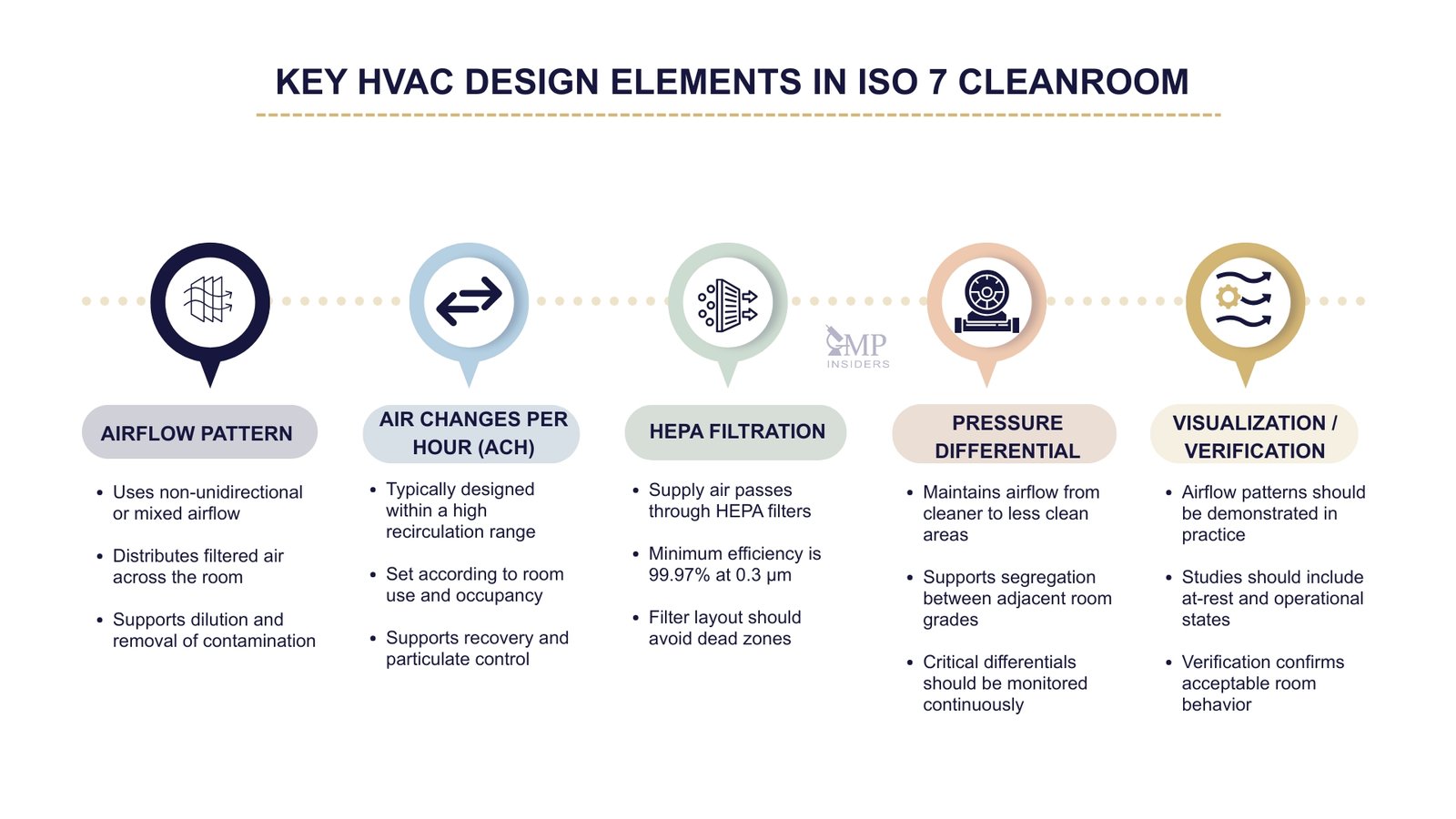
Airflow Type in Class ISO 7: Turbulent/Mixed Flow
Class ISO 7 environments rely on non-unidirectional airflow, which distributes clean air throughout the room via supply diffusers and removes it through wall or ceiling-mounted returns.
Key design expectations include:
- supply of appropriately filtered air to the cleanroom,
- air distribution that supports effective flushing of the room,
- extraction arranged to minimize stagnation and uncontrolled recirculation,
- airflow patterns that do not permit ingress from lower-grade to higher-grade areas,
- airflow behavior that remains acceptable during routine operator movement and process activity
| Design element | Expected design principle | Practical concern if poorly controlled |
|---|---|---|
| Filtered air supply | Clean air should enter the room through a suitable filtered supply system | Inadequate particle removal and unstable classification |
| Room flushing | Air distribution should support dilution of contamination throughout the space | Local contamination build-up or poor recovery |
| Return placement | Returns should be arranged to support effective removal of room air | Stagnation or uncontrolled recirculation |
| Flow direction | Airflow should not allow ingress from lower-grade to higher-grade areas | Increased contamination risk to adjacent critical zones |
| Operational robustness | Airflow should remain acceptable during routine movement and process activity | Loss of control during actual use rather than only at rest |
For rooms that directly interface with Grade A or Grade B operations, airflow performance should be considered in relation to the wider contamination control strategy rather than as an isolated room parameter. The question is not only whether the room achieves ISO 7 classification, but whether the airflow pattern continues to support the protection of the critical zone during actual use.
Air Changes per Hour (ACH) for ISO 7 Cleanrooms
Neither ISO 14644-1 nor Annex 1 assigns a fixed air change rate for ISO 7 cleanrooms. Air change rates should therefore be established through design qualification and demonstrated to be sufficient for the intended room function, occupancy, equipment load, heat gain, and recovery requirements.
In practice, many ISO 7 cleanrooms are designed with relatively high recirculation rates, but the regulatory expectation is performance-based rather than prescriptive.
Typical ISO 7 ACH range:
- 35–60 ACH (industry-common ranges)
- Higher when:
- room volume is large
- occupancy is high
- equipment generates heat or particles
- the area directly supports Grade A operations
From an operational perspective, the ventilation rate should be sufficient to support:
- maintenance of the required particulate classification,
- timely recovery following personnel activity or operational disturbance,
- control of temperature and relative humidity where these are process-relevant,
- sustained environmental stability during routine occupancy,
- protection against ingress of contamination from adjacent lower-grade spaces.
When numerical ACH ranges are used in design documents or internal standards, they should be presented as engineering setpoints or typical design values, not as fixed regulatory limits for ISO 7 classification.
HEPA Filtration Requirements for Class ISO 7
ISO 7 cleanrooms require air supplied through HEPA filters with a minimum efficiency of 99.97% at 0.3 μm.
HEPA considerations in ISO 7:
- Filters may be terminal (ceiling-mounted) or part of ducted FFUs.
- Layout must avoid dead zones or uneven airflow distribution.
- Filters must undergo integrity testing during qualification and periodically (typically annually).
- Filter leaks directly affect ISO 5 zones downstream.
HEPA filtration is foundational to achieving ISO 7 classification, even though airflow is not unidirectional.
See Also: Types of HEPA Filters Used in Pharmaceutical Industry
Pressure Cascades and Room Pressurization
Pressure differentials remain a critical HVAC control in ISO 7 cleanrooms, particularly when these rooms serve as the background for higher-classified areas.
Annex 1 states that cleanrooms should be supplied with filtered air that maintains a positive pressure and/or an airflow relative to the background environment of a lower grade under all operational conditions, and that adjacent rooms of different grades should have an air pressure difference of a minimum of 10 Pa as a guidance value.
FDA guidance similarly recommends at least 10–15 Pa between rooms of different classifications.
In practical terms, this means:
- The pressure cascade should direct air from cleaner to less clean areas,
- Pressure set points should be defined according to the room function and process risk,
- Pressure differences identified as critical should be continuously monitored and recorded,
- Warning or alarm systems should indicate loss of air supply or reduction below established limits,
- Any pressure reversal or unexplained loss of differential should be assessed for potential product impact.
For that reason, it is more accurate to describe 10 Pa minimum as the Annex 1 guidance value, while noting that some facilities operate with higher nominal set points, such as 10–15 Pa, to maintain a reliable cascade within their operating tolerances.
Airflow Visualization and HVAC Verification
Airflow visualization studies are an important part of demonstrating that the HVAC design functions as intended. Annex 1 requires airflow patterns within cleanrooms and zones to be visualized to show that there is no ingress from lower-grade to higher-grade areas and that air does not travel from less clean areas, operators, or equipment toward higher-risk zones.
These studies should be performed both at rest and in operation, including, where relevant, the simulation of operator interventions, and the recordings should be retained.
For ISO 7 cleanrooms, airflow visualization should typically confirm:
- absence of reverse flow from lower-grade adjacent areas,
- acceptable air movement at transfer interfaces and pass-through points,
- no persistent stagnation behind large equipment or fixed installations,
- no airflow pattern that adversely affects adjacent Grade A or Grade B protection,
- acceptable airflow behavior during representative personnel movement and operating conditions.
Where unidirectional airflow is used at critical locations, Annex 1 also requires that airflow-visualization studies be correlated with air-speed measurements.
Impact of Personnel and Equipment
The operational challenge in ISO 7 cleanrooms is often greater than in more tightly localized critical zones because these rooms accommodate greater movement, more interventions, and greater variation in heat and airflow disturbances.
Operator traffic, material handling, equipment geometry, and thermal loads can all influence room behavior and should therefore be considered during design qualification, airflow studies, and routine reassessment.
Particular attention should be given to the following:
- personnel movement as a source of particle generation and local airflow disturbance,
- gowning and transfer activities near entry points, hatches, and airlocks,
- heat-generating equipment that may create convection currents,
- equipment placement that obstructs supply or extract pathways,
- changes in room layout or process configuration that may alter established airflow patterns.
| Disturbance source | Typical effect on room behavior | Why it should be considered |
|---|---|---|
| Personnel movement | Generates particles and disrupts local airflow | Can reduce room stability during routine use |
| Gowning and transfer activity | Disturbs airflow near doors, hatches, and airlocks | May increase contamination movement at interfaces |
| Heat-generating equipment | Creates convection currents | Can alter established airflow behavior |
| Poor equipment placement | Obstructs supply or return pathways | Can create dead zones or uneven flushing |
| Layout or process changes | Changes airflow patterns within the room | May invalidate previous airflow assumptions or study results |
Overall, HVAC design for ISO 7 should not be approached as a simplified version of either ISO 8 comfort ventilation or ISO 5 critical zone protection. It is a controlled engineering system that must maintain classification, preserve pressure-cascade integrity, and support the contamination-control function of the surrounding sterile manufacturing environment.
Class ISO 7 Layout and Room Design
The layout of an ISO 7 cleanroom is a critical determinant of its ability to maintain a controlled state during operation. This is particularly important where the room supports higher-classified aseptic activities, since the arrangement of spaces, equipment, personnel routes, and transfer interfaces can directly influence airflow behavior, cleanliness, and contamination risk.
In this context, room design should not be viewed solely as a matter of convenience or workflow. It is an integral part of the contamination control strategy and should support the intended state of control under both at-rest and operational conditions.
Unlike ISO 5 zones, which are designed to protect exposed products and critical surfaces through first-air protection, ISO 7 rooms typically serve as supporting environments. Their design must therefore accommodate routine activity while still controlling the effects of operator movement, material transfer, equipment placement, and environmental disturbance.
The objective is to achieve a layout that supports predictable flow, minimizes unnecessary interventions, and reduces opportunities for contamination ingress or redistribution.
The Role of ISO 7 in a GMP Cleanroom Suite
ISO 7 cleanrooms serve as both processing and staging areas. They are more controlled than ISO 8 rooms but more flexible than ISO 5, making them the central zone for preparation before entering aseptic operations.
Class ISO 7 typically functions as:
- The background environment for RABS, isolators, and laminar-flow ISO 5 units
- The buffer zone between ISO 8 (preparation) and ISO 5 (critical processing)
- A staging area for sterile components before transfer to ISO 5
- A gowning area for Grade B personnel gowning
- A corridor or transfer space between clean areas of differing grades
Its position in the cleanroom hierarchy makes layout decisions especially important.
Material Flow Through ISO 7 Cleanrooms
The transfer of materials and equipment into and out of cleanrooms is recognized as a major potential source of contamination. Accordingly, the layout of an ISO 7 room should support a controlled and, where required, unidirectional material flow. Incoming and outgoing items should not compete for the same pathway, as this may compromise cleanliness or create avoidable operational disturbance.

Annex 1 states specifically that transfer into Grade A and B areas should occur via a unidirectional process and that, where this is not possible, time-based separation and additional controls should be applied.
Good layout practice for material flow includes:
- defined routes for incoming and outgoing materials,
- transfer points positioned to minimize cross-traffic and unnecessary door openings,
- use of material airlocks or pass-through hatches where movement occurs between grades,
- pre-cleaning or disinfection of items before entry, in line with the contamination control strategy,
- minimization of in-room storage, particularly where stored items may obstruct airflow or hinder cleaning.
It is also better to avoid broad wording such as “material flow must be unidirectional” for all ISO 7 rooms. That concept is specifically critical for transfer into Grade A and Grade B areas; in other ISO 7 applications, the design requirement is a controlled, justified, and contamination-minimizing flow rather than a universal one-way rule.
Personnel Flow and Gowning Requirements
Personnel movement should be considered one of the main sources of contamination within ISO 7 environments. The room layout should therefore limit unnecessary entry, exit, crossing, and backtracking.

FDA guidance explicitly states that both personnel and material flows should be optimized to prevent unnecessary activities that could increase the risk of contamination, and Annex 1 requires that movement in clean areas be slow, controlled, and methodical.
Typical gowning requirements for ISO 7/Grade B at-rest include:
- Sterile hood
- Cleanroom hair cover
- Sterile face mask
- Sterile coverall
- Sterile gloves
- Sterile, low-lint boot covers
From a layout perspective, this means that personnel routes should be clear and logical, with movement patterns that support the sequence of gowning, entry, operation, and exit. Appropriate design features include:
- personnel airlocks arranged in increasing order of cleanliness,
- separation of personnel and material airlocks wherever possible,
- reduced need for repeated entry and exit during operations,
- room arrangement that avoids unnecessary movement close to critical interfaces,
- sufficient space for gowning, checks, and controlled movement without crowding.
With respect to gowning, it is important not to present a single fixed gowning set as though it applies to every ISO 7 room. Gowning is determined by the assigned GMP grade and the activity performed, not by ISO class alone.
Where an ISO 7 room functions as a Grade B background, Annex 1 requires every operator entering Grade A or B areas to gown in clean, sterilized protective garments, including eye coverings and masks, with sterile footwear and gloves managed under defined controls. Gloves should be disinfected regularly during operations, and damaged garments or gloves should be replaced immediately.
Pass Boxes and Transfer Systems
Pass boxes, material airlocks, and similar transfer interfaces are important control points in ISO 7 design because they connect areas of different cleanliness grades and can disrupt pressure and airflow if poorly designed.
Annex 1 requires airlocks to provide physical separation and minimize microbial and particle contamination between areas. It also states that pass-through hatches should be designed to protect the higher-grade environment, for example, through effective flushing with filtered air.
Related Article: Types of Pass Boxes Used in GMP
Critical design elements:
- Interlocked doors to protect pressure cascade
- Smooth, easy-to-clean internal surfaces
- Defined sanitization methods (e.g., spraying, wiping, VHP if applicable)
- Proper sizing to avoid door obstruction or overcrowding
- External indicators to show door lock status
Pass boxes reduce traffic through cleanroom doors and help maintain environmental stability.
Surface and Material Selection
Internal surfaces in ISO 7 rooms should support effective cleaning and should not contribute to particle generation or microbial retention.
Annex 1 states that exposed surfaces in cleanrooms should be smooth, impervious, and unbroken, and that room materials should be selected to minimize particle generation and to withstand repeated application of cleaning, disinfectant, and sporicidal agents where used. It also states that recesses difficult to clean should be avoided and that projecting ledges, shelves, cupboards, and similar features should be kept to a minimum.
Suitable construction and finish principles, therefore, include:
- smooth, sealed wall and ceiling systems,
- flush-mounted fixtures where possible,
- sealed penetrations around pipes, ducts, and cables,
- flooring systems that are seamless or effectively sealed at joints,
- observation panels and glazing installed flush and sealed to the surrounding structure.
| Surface / design feature | Preferred characteristic | Reason |
|---|---|---|
| Walls and ceilings | Smooth, sealed, impervious, and unbroken | Supports effective cleaning and minimizes particle retention |
| Fixtures and fittings | Flush-mounted where possible | Reduces ledges, recesses, and difficult-to-clean areas |
| Penetrations | Fully sealed around ducts, pipes, and cables | Prevents leakage paths and dirt accumulation |
| Flooring | Seamless or effectively sealed at joints | Improves cleanability and reduces contamination retention |
| Observation panels / glazing | Flush and sealed to surrounding structure | Maintains cleanable surfaces and room integrity |
| Construction materials | Non-shedding, cleanable, and chemical-resistant | Supports long-term use under GMP cleaning conditions |
Materials commonly used in practice include stainless steel, coated metals, cleanroom-grade composite wall systems, resin-based flooring, and sealed glass viewing panels. However, the regulatory requirement is performance-based: the material must be non-shedding, cleanable, resistant to the chemicals used, and suitable for the room’s intended operation.
One additional point that is often missed: if an ISO 7 room is assigned as Grade B, sinks and drains are prohibited there under Annex 1. In lower-grade cleanrooms, where drains are permitted, they require appropriate design controls, such as air breaks, traps, or water seals, as well as routine cleaning and disinfection.
Room Zoning and Functional Segmentation
ISO 7 rooms perform best when different activities are clearly segregated within the space or across connected spaces. Combining staging, gowning, equipment preparation, transfer, and operational support within a single undifferentiated footprint increases traffic, operational interference, and the risk of disrupting established airflow patterns. FDA guidance also emphasizes equipment layout and ergonomics as part of reducing contamination risk.
Depending on the process, functional segregation may include:
- a preparation or staging area,
- a defined transfer interface zone,
- a personnel transition or final preparation area,
- an operational support zone adjacent to the aseptic core,
- separated ingress and egress routes where justified by risk.
The intent of zoning is to ensure that the layout supports the required airflow, cleaning access, monitoring coverage, and discipline of movement associated with each activity.
Lighting and Visibility Considerations
Lighting should support operations, inspections, cleaning, and environmental monitoring without creating surfaces that are difficult to clean or features that disrupt airflow. Although Annex 1 is not highly prescriptive on lighting design, the same cleanability and contamination-control principles apply to light fittings as to other room elements. In practical terms, luminaires should be integrated so that they do not create inaccessible ledges, recesses, or leakage paths above the ceiling void.
Appropriate design features generally include:
- flush-mounted or sealed fixtures,
- smooth external surfaces,
- installation that avoids dust traps and difficult-to-clean housings,
- sufficient illumination for safe operation and visual inspection.
| Design aspect | Recommended approach | Why it matters |
|---|---|---|
| Fixture installation | Use flush-mounted or sealed luminaires | Prevents ledges and difficult-to-clean housings |
| External finish | Keep surfaces smooth and cleanable | Reduces dust traps and contamination retention |
| Ceiling integration | Avoid leakage paths or inaccessible recesses above the ceiling void | Supports room integrity and cleanability |
| Illumination level | Provide sufficient lighting for operation, inspection, and cleaning | Supports safe and accurate work |
| Airflow compatibility | Ensure lighting design does not interfere with room airflow behavior | Helps preserve environmental control |
Cleaning and Disinfection Requirements for ISO 7 Cleanrooms
Cleaning and disinfection in ISO 7 cleanrooms should be established as part of the site contamination control strategy and should be sufficient to maintain the room in a controlled state during routine operation.

Where an ISO 7 room serves as the surrounding environment for higher-classified aseptic operations, the effectiveness of the cleaning program is directly relevant to both particulate and microbiological control. In this context, cleaning should not be treated as a housekeeping activity alone, but as a documented control measure that supports the area’s qualified status and the protection of adjacent critical zones.
The cleaning strategy should reflect the room’s actual use, including personnel occupancy, equipment configuration, material transfer, and the assigned GMP grade. For that reason, fixed generic statements such as “ISO 7 requires less intensive cleaning than ISO 5” are not sufficiently precise for GMP use.
The required program depends on process risk and room function, particularly where the same ISO class may be used in different GMP contexts.
Cleaning Principles for ISO 7 Cleanrooms
For ISO 7 cleanrooms, the cleaning program should be written, approved, and performed in accordance with defined methods and frequencies. Annex 1 is explicit that cleanrooms should be cleaned and disinfected thoroughly in accordance with a written program, and that prior cleaning is necessary for disinfection to be effective.
Core principles include:
- Visible soil and surface residues should be removed before disinfectant application
- The program should define the cleaning method, sequence, contact time, and frequency for each area and surface type,
- Cleaning should also address the removal of disinfectant residues, not only microbial reduction,
- The cleaning technique and materials used should minimize particle generation and should not adversely affect the cleanroom environment,
- Activities should be documented at the time they are performed, in line with GMP expectations for traceability and control.
Where isolators, RABS interfaces, transfer hatches, or other local high-risk features are present within or adjacent to the ISO 7 area, the cleaning program should also address those interfaces specifically, since residues or ineffective disinfection at such points may compromise the wider cleanroom state of control.
Selection and Validation of Disinfectants
Disinfectants and detergents used in cleanrooms should be selected based on demonstrated suitability for their intended use. Annex 1 requires the disinfection process to be validated and states that validation studies should demonstrate the suitability and effectiveness of the disinfectants in the specific manner in which they are used and on the relevant surface materials. Validation should also support the in-use expiry period of prepared solutions.
A suitable disinfectant program should therefore address:
- activity against the facility flora likely to be encountered in the area,
- suitability for the surface types present in the room,
- defined contact time and application method,
- compatibility with repeated use on cleanroom finishes and equipment exteriors,
- stability and in-use hold time of prepared or diluted solutions,
- effectiveness of the program when used in conjunction with prior cleaning.
| Selection / validation factor | What should be demonstrated |
|---|---|
| Activity against likely facility flora | The disinfectant is suitable for the microorganisms expected in the area |
| Surface compatibility | The disinfectant can be used repeatedly on room finishes and equipment exteriors without damage |
| Contact time | The required exposure time is defined and achievable in routine use |
| Application method | The disinfectant is effective in the way it is actually applied in the cleanroom |
| Stability / in-use hold time | Prepared or diluted solutions remain suitable throughout their defined in-use period |
| Use with prior cleaning | The program is effective when combined with the required cleaning step before disinfection |
| Sterility requirement | Sterile disinfectants / detergents are used where required by the assigned GMP grade |
Annex 1 states that disinfectants and detergents used in Grade A and Grade B areas should be sterile prior to use, while disinfectants used in Grade C and D may also need to be sterile, as determined in the contamination control strategy. Accordingly, if an ISO 7 room is used as a Grade B background, sterile disinfectants and detergents are expected.
Typical disinfectants in ISO 7:
- 70% IPA or ethanol (frequent use)
- Quaternary ammonium compounds
- Hydrogen peroxide blends
- Chlorine-based agents (periodic)
- Sporicidal disinfectants (weekly or monthly)
More than one type of disinfectant should be used when different modes of action are needed to ensure effectiveness against bacteria and fungi, and the disinfection program should include periodic use of a sporicidal agent. Routine monitoring is then used to assess effectiveness and detect changes in microbial flora, including possible resistance to the regimen in use.
Cleaning Frequency in Class ISO 7
Cleaning frequency should be defined through quality risk management and should reflect room use, occupancy, environmental monitoring data, and the types of operations performed. Neither Annex 1 nor FDA guidance provides a universal ISO 7 cleaning timetable that can be applied without justification.
In practice, the program commonly includes a combination of the following:
- routine cleaning before the start of operations, where applicable,
- cleaning and disinfection of work surfaces and frequently touched items during or after activities that increase contamination risk,
- end-of-operation or end-of-shift cleaning of accessible room surfaces and equipment exteriors,
- periodic application of a sporicidal agent according to a defined written schedule,
- additional cleaning after spills, maintenance, engineering work, interventions, or environmental monitoring excursions.
Where the area directly supports aseptic processing, the cleaning frequency should be sufficient to maintain the required state of control throughout the campaign or operating period, not merely to restore the room at the end of the day.
Cleaning of Critical and High-Traffic Surfaces
Within ISO 7 cleanrooms, certain surfaces generally require particular attention because they are more frequently contacted by personnel or are located at key transfer points. The cleaning program should explicitly identify such surfaces rather than rely solely on broad room-based instructions.
These commonly include:
- door handles and push plates,
- pass-through hatch handles and transfer interfaces,
- benches, staging surfaces, and preparation tables,
- outer surfaces of equipment located within the room,
- controls, interlock buttons, and operator touchpoints,
- floor areas near entrances, material transfer locations, and operational routes.
| Surface / area | Why it needs specific attention |
|---|---|
| Door handles and push plates | High-touch points with frequent operator contact |
| Pass-through hatch handles and transfer interfaces | High-risk locations for contamination during transfer between areas |
| Benches, staging surfaces, and preparation tables | Repeated use during routine cleanroom activities |
| Outer surfaces of room equipment | Frequently exposed during operations and cleaning |
| Controls, interlock buttons, and operator touchpoints | Recurrent personnel contact may increase contamination risk |
| Floor areas near entrances and transfer routes | More likely to be affected by traffic and movement |
Where environmental monitoring identifies recurring contamination at specific locations, those sites should be reassessed from both a cleaning-frequency perspective and a layout, operator practice, and surface-design standpoint.
Cleaning Tools and Materials Approved for ISO 7
Cleaning tools and materials used in ISO 7 areas should be suitable for cleanroom use and compatible with the disinfectants employed. Annex 1 states that materials used within cleanrooms should be selected to minimize particle generation and to permit repeated application of cleaning, disinfectant, and sporicidal agents, where used.
Accordingly, the program should typically use:
- low-lint wipes and mop materials suitable for classified areas,
- cleaning tools constructed from non-shedding, cleanable materials,
- containers that are cleaned and sterilized where applicable, before use,
- pre-prepared or qualified diluted disinfectants held only for their defined in-use period,
- application methods that are controlled and reproducible.
Qualification and Requalification of ISO 7 Cleanrooms
ISO 7 cleanrooms should be qualified to demonstrate their ability to maintain the required classified state for their intended use. In sterile manufacturing, this is particularly important when the room supports higher-grade operations, since cleanroom qualification provides documented evidence that the HVAC system, room configuration, airflow pattern, and environmental controls operate together to support contamination control.
Under Annex 1, cleanroom qualification, including classification, should be clearly distinguished from routine environmental monitoring.
Qualification of ISO 7 areas should typically include, where relevant:
- cleanroom classification by total particle concentration,
- integrity testing of final filters,
- airflow volume measurement,
- air pressure differential verification,
- airflow direction testing and airflow visualization,
- microbial airborne and surface assessment,
- temperature and relative humidity verification,
- recovery testing,
- containment leak testing, where applicable.
Requalification should be performed at defined intervals and after changes that may affect cleanroom performance, such as HVAC modification, major maintenance, layout change, or repeated adverse monitoring trends.
Under Annex 1, the maximum requalification interval is 6 months for Grade A and Grade B areas and 12 months for Grade C and Grade D areas. Because ISO 7 may be used in different GMP contexts, the applicable requalification frequency should be linked to the assigned cleanroom grade rather than to ISO class alone.
Related Article: Cleanroom Qualification and Validation in GMP
FAQ
Can ISO 7 Be Used for Open Aseptic Processing on Its Own?
No. ISO 7 should not be presented as sufficient on its own for operations in which sterile products, sterile components, or critical product-contact surfaces are exposed. Under EU GMP Annex 1, aseptic compounding, aseptic mixing, exposed aseptic connections, and filling activities are performed in Grade A, typically with a Grade B background. ISO 7 may serve as part of that supporting background function, but it is not a substitute for the local Grade A conditions required at the critical zone.
Does ISO 7 Always Correspond to Grade B?
No. ISO classification and GMP grading are related but not interchangeable. The assigned GMP grade depends on the operation performed, the room function, and the state in which the area is assessed. In practice, an ISO 7 room may support various activities and should not automatically be classified as Grade B in all cases.
How Much Personnel Activity Is Acceptable in ISO 7?
Not in every case. The requirement depends on the GMP function of the airlock or transfer interface. Annex 1 states that entry and exit doors should not be opened simultaneously, and that interlocking systems should be used for airlocks leading to Grade A and Grade B areas. For airlocks leading to Grade C and Grade D areas, a visual and/or audible warning system is the minimum expectation. Where necessary to maintain segregation, a time delay between door closure and opening should also be established.
Is Continuous Monitoring Required in ISO 7?
If the ISO 7 room functions as a Grade B background, Annex 1 recommends a monitoring approach similar to Grade A for non-viable particles, although at a reduced sample frequency. If the room supports a lower-grade operation, the monitoring approach may be based more directly on quality risk management, room use, and proximity to critical zones. For that reason, routine monitoring expectations should be linked to the assigned GMP grade and process risk, not to ISO 7 alone.
How Long Is the Typical Recovery Time for ISO 7 Rooms?
Recovery times vary depending on ACH and room volume, but typically range from 5–15 minutes after a disturbance. Shorter recovery times indicate efficient airflow and good diffuser/return design. Longer times can signal HVAC imbalance, obstructions, or low supply flow. Recovery time is verified during qualification and revisited if major layout or HVAC changes occur. Poor recovery is a common cause of EM instability.
Can ISO 7 be Downgraded to ISO 8 If Operations Change?
Yes, but only after reassessment and requalification. Downgrading requires adjustments to HVAC, monitoring plans, cleaning frequency, and gowning. Regulatory expectations must be considered, especially if the area previously supported aseptic operations. The contamination control strategy must be updated to reflect the new classification. Downgrading is less complex than upgrading, but it still must follow change control.
Final Thoughts
ISO 7 cleanrooms provide the controlled background necessary to support higher-classified operations and play a direct role in protecting adjacent ISO 5 or Grade A critical zones. Their condition, therefore, significantly influences the overall effectiveness of contamination control within the facility.
The importance of ISO 7 should not be assessed only by its classification, but by its function within the wider cleanroom system. Airflow performance, pressure cascade stability, personnel and material movement, cleaning and disinfection, and environmental monitoring all contribute to determining whether the area remains under control. Where weaknesses arise in these supporting conditions, the effect is often seen first in the background environment before it becomes evident in more critical zones.
For that reason, ISO 7 operation requires more than initial qualification alone. Continued control depends on consistent application of defined procedures, effective oversight, and a cleanroom design that remains suitable for the activities performed. When properly designed, qualified, and routinely managed, ISO 7 cleanrooms provide the stable supporting environment needed to sustain aseptic operations and reinforce the wider contamination control strategy.














